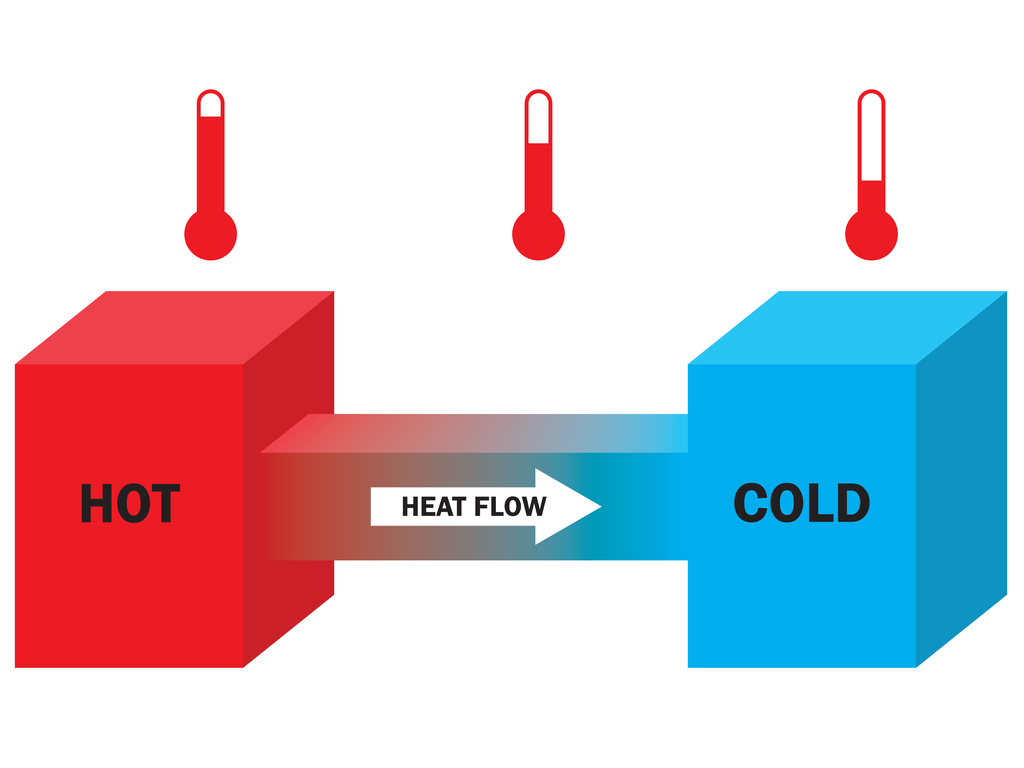
Why Does Heat Transfer From Hot To Cold
Ever wondered why that perfectly brewed cup of coffee gets lukewarm after a while? Or why your ice cream melts faster on a summer's day than in your freezer? It all boils down (pun intended!) to a fundamental principle of physics: heat transfer. But why does heat always flow from hot to cold? Let’s dive into this intriguing phenomenon in a way that’s easier to digest than a slice of key lime pie.
The Molecular Dance: A Kinetic Perspective
Imagine a bustling dance floor. On one side, we have a group of energetic dancers (hot molecules) boogying with gusto, bumping into each other and spreading their energy. On the other side, we have a more relaxed crowd (cold molecules) taking it easy. When these two groups mingle, what happens? The energetic dancers inevitably start bumping into the less energetic ones, transferring some of their exuberance. This, in a nutshell, is how heat transfer works at a molecular level.
Specifically, heat is a form of kinetic energy – the energy of motion. Hotter substances have molecules moving faster than cooler substances. When these two substances come into contact, the faster-moving molecules collide with the slower-moving ones, transferring some of their kinetic energy. This transfer continues until both substances reach thermal equilibrium, meaning they have the same temperature. Think of it like equalizing the energy on the dance floor so everyone's moving at a similar pace.
Must Read
Entropy: The Universe’s Natural Inclination Towards Disorder
Okay, so molecules are dancing. But why only from hot to cold? The answer lies in a concept called entropy. Entropy, in simple terms, is a measure of disorder or randomness in a system. The universe tends to move toward a state of higher entropy. Think of it like this: it's easier to mess up your room than to perfectly organize it. Disorder is the natural state.
When heat flows from hot to cold, it's increasing the overall entropy of the system. The energy is spreading out and becoming more evenly distributed, creating a more disordered state. The opposite – heat flowing from cold to hot – would require a decrease in entropy, which the universe isn't too keen on. It's like trying to force all the dancers to stand perfectly still – against their nature!

Practical Tips and Cultural Connections
Understanding heat transfer isn't just for physicists; it has practical applications in our daily lives. Here are a few examples:
- Insulating your home: Insulation works by slowing down the rate of heat transfer. It creates a barrier that prevents heat from escaping in the winter and entering in the summer, keeping your home comfortable and saving you money on energy bills.
- Cooking: Think about using a cast iron skillet. It heats up evenly and retains heat well, ensuring your food cooks consistently. This is thanks to its high thermal conductivity and heat capacity.
- Clothing: Wearing layers in cold weather traps air between the layers, which acts as an insulator. This prevents your body heat from escaping.
Culturally, different societies have developed ingenious ways to manage heat. For example, the architecture of many Mediterranean countries features thick walls and small windows to keep buildings cool during the hot summer months. Similarly, the nomadic Tuareg people of the Sahara wear loose-fitting, dark-colored clothing to protect themselves from the scorching sun. The dark color absorbs more heat, but the loose fit allows for ventilation, keeping them cooler than if they wore tight-fitting, light-colored clothes. These are examples of applied thermodynamics in action!

Fun Fact!
Did you know that a thermos flask, also known as a vacuum flask, works by minimizing all three modes of heat transfer: conduction, convection, and radiation? The vacuum between the inner and outer walls prevents heat transfer by conduction and convection, while the reflective surfaces minimize heat transfer by radiation. Pretty cool, huh?
Reflection
The principle of heat transfer – the seemingly simple flow from hot to cold – underscores a fundamental truth about the universe: energy is constantly seeking equilibrium. This constant flow, this natural inclination towards disorder, is a reminder that everything is interconnected and that even the smallest interactions can have far-reaching consequences. Just like the energetic dancers spreading their exuberance, we too are constantly influencing the world around us, whether we realize it or not. So next time you see ice melting in your glass, remember the dancing molecules and the grand, ever-unfolding story of the universe seeking balance. It's a good reason to pause, appreciate, and maybe even learn something new!