In What Form Is Energy Stored In Batteries

Isn't it amazing how we can carry pockets of power with us everywhere? From smartphones buzzing with notifications to electric cars silently gliding down the street, batteries have become an indispensable part of modern life. We rely on them so heavily, but have you ever stopped to think about how these little boxes (or sometimes not-so-little boxes!) actually store energy?
The beauty of batteries lies in their ability to provide on-demand power. Imagine a world without them. Our phones would be tethered to walls, and laptops would be stationary dinosaurs. Batteries provide freedom and convenience, allowing us to power our lives untethered. They’re crucial for everything from hearing aids giving someone the gift of sound, to pacemakers keeping hearts beating steadily, to emergency backup power ensuring hospitals can function during outages.
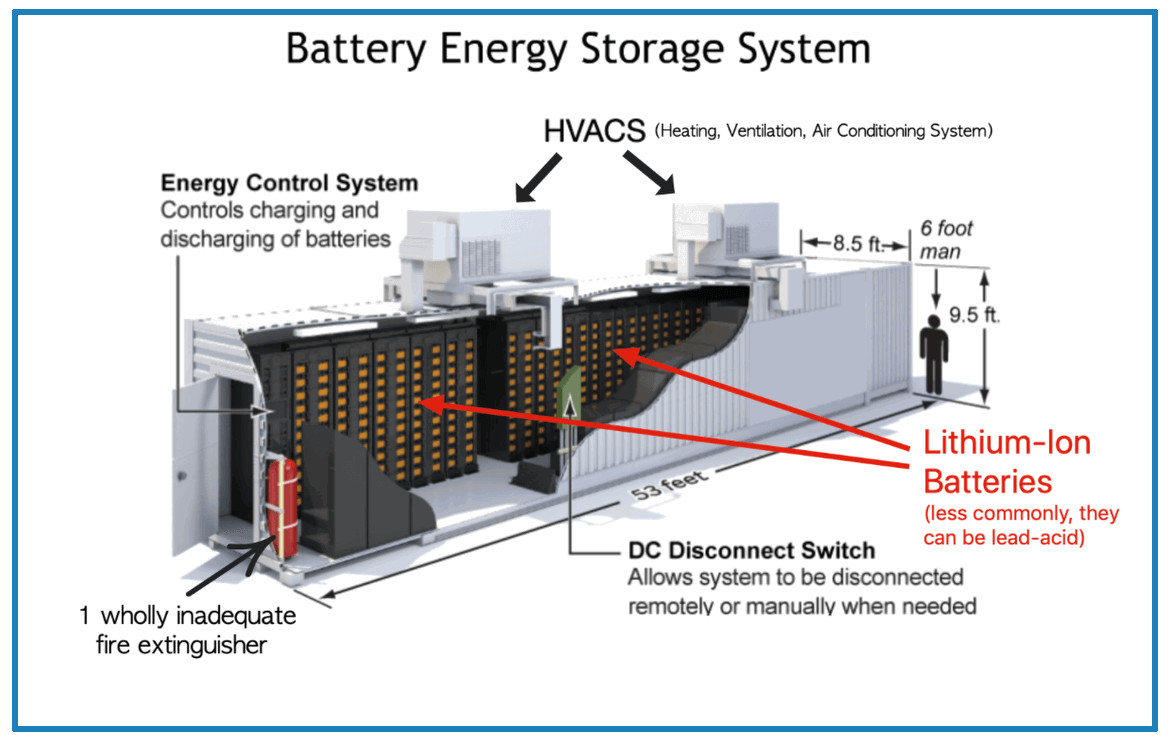
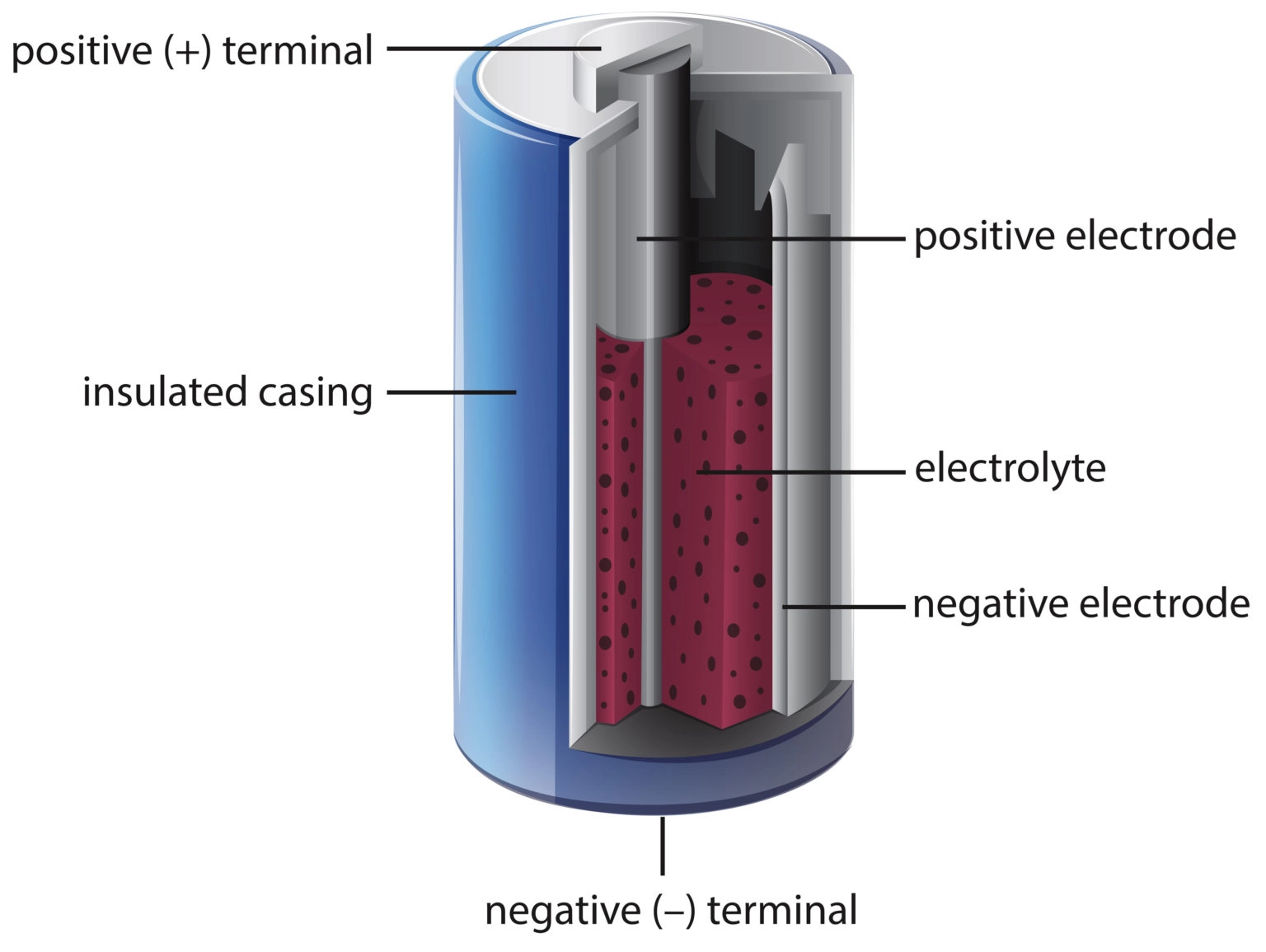
So, the big question: What form does this stored energy take? It's not like they're filled with miniature lightning bolts! The answer lies in the fascinating world of chemistry. Batteries store energy in the form of chemical potential energy. Think of it as a carefully arranged set of chemicals, eager to react and release energy. Inside a battery, you'll find two electrodes – a cathode (positive end) and an anode (negative end) – submerged in an electrolyte, which facilitates the movement of ions.
Must Read
When you connect a device to a battery, a chemical reaction kicks off. Electrons flow from the anode to the cathode through the external circuit, powering your device. This flow creates an electrical current. As the reaction proceeds, the chemicals are gradually used up, and the battery discharges. Different types of batteries, like lithium-ion (found in most smartphones), alkaline (common in remote controls), and lead-acid (used in cars), use different chemical reactions, but the fundamental principle remains the same: chemical energy transformed into electrical energy.
Here are a few practical tips to make your battery experiences even better:
- Avoid extreme temperatures: Heat and cold can significantly impact battery life. Don't leave your phone baking in the sun or freezing in your car.
- Use the right charger: Using a charger not specifically designed for your device can damage the battery. Always stick to the manufacturer's recommended charger.
- Partial charging is often better: Unlike older battery technologies, lithium-ion batteries generally prefer being charged in shorter bursts rather than being fully discharged and then charged to 100%.
- Store batteries properly: If you're storing batteries for a long time, keep them in a cool, dry place. Remove them from devices you're not using to prevent corrosion.
- Consider battery recycling: Batteries contain valuable materials that can be recovered and reused. Look for local recycling programs to dispose of them responsibly.
Understanding the science behind batteries might not seem immediately exciting, but it allows us to appreciate the ingenuity and complexity packed into these everyday devices. Next time you reach for your phone or start your car, remember the intricate dance of chemicals happening inside that battery, silently powering your world.