How Many Micrograms Are In A Milliliter

Okay, so picture this: I'm trying to bake this ridiculously complicated cake I saw on, like, a Martha Stewart rerun. It calls for, and I quote, "300 micrograms of vanilla extract." I'm staring at my measuring spoons, my graduated cylinder... all these lovely things calibrated in milliliters. Panic. Ensues. Like, seriously, who measures vanilla in micrograms? Probably Martha, I guess. But the point is, that's when I realized I really needed to understand the whole micrograms-to-milliliters conversion thing. (Because, let's be honest, I was NOT about to eyeball it with my expensive vanilla!)
So, let’s dive into this micro-mystery! Basically, we're talking about converting between units of mass (micrograms) and units of volume (milliliters). But here's the twist: it all depends on the density of the substance you're measuring.
Think of it this way: a milliliter of feathers is going to weigh a lot less than a milliliter of, say, liquid mercury. (Please don’t play around with mercury, by the way. Just a hypothetical example!) So, just knowing the volume isn't enough.
Must Read
The Magic Formula (Sort Of)
Here's where things get slightly... mathematical. The formula that ties everything together is:
Density = Mass / Volume
Which can be rearranged to find the mass:
Mass = Density * Volume
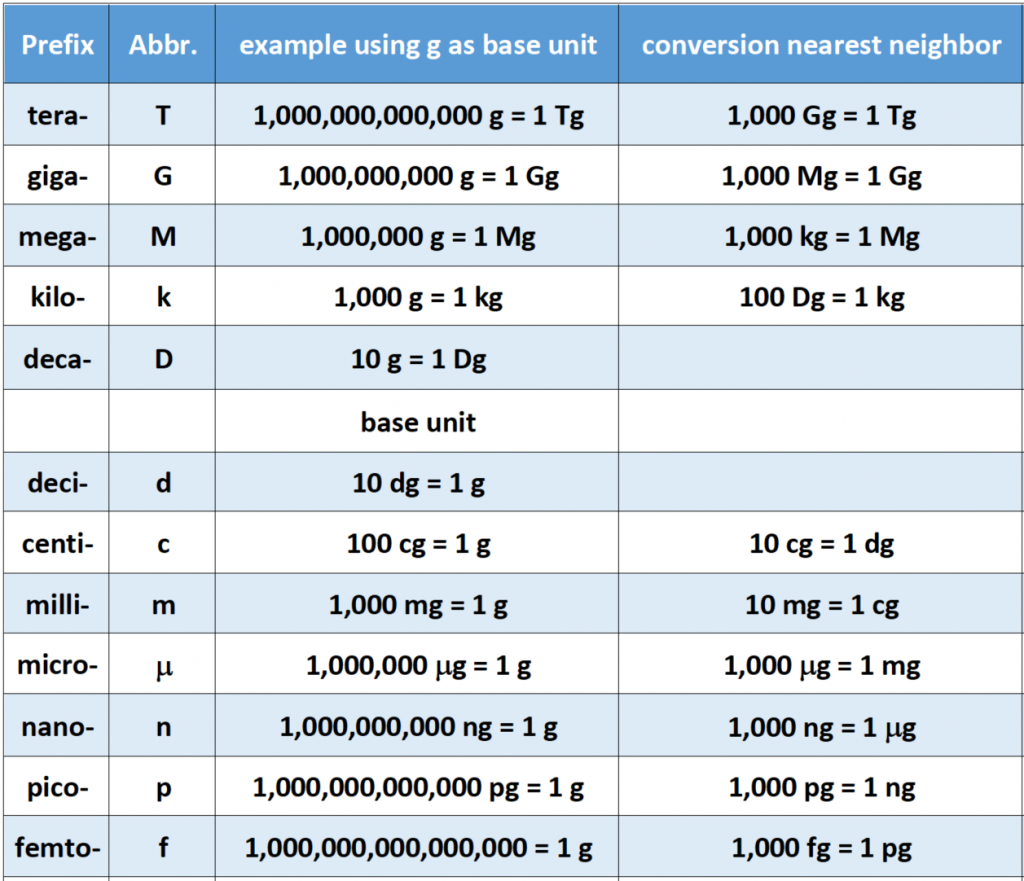
Okay, okay, I know. Math. But stick with me! We want to find the mass in micrograms when we know the volume in milliliters. So, we need to make sure our units are consistent. Since 1 milliliter (mL) is also equal to 1 cubic centimeter (cm3), that's easy!
The most important thing is that the density needs to be in micrograms per milliliter (µg/mL). Once you have that, the conversion is simple. You just multiply the volume (in mL) by the density (in µg/mL) to get the mass in micrograms (µg).
Easy peasy, right? (Don't answer that if you're still confused. Keep reading!)
Why Density is King (or Queen)
Let's say you're working with water. The density of water is approximately 1 gram per milliliter (1 g/mL). That's great, but we need micrograms! So, let's convert.
Since 1 gram (g) is equal to 1,000,000 micrograms (µg), the density of water is approximately 1,000,000 µg/mL.
Therefore, if you have 1 mL of water, it weighs approximately 1,000,000 micrograms. That's a million!
But here's the kicker: this only applies to water. What if you’re dealing with something else, like an oil, a solvent, or, yes, even vanilla extract?
You need to know the density of that specific substance. You can usually find this information on the product label, the manufacturer's website, or in a material safety data sheet (MSDS). Don't just assume it's the same as water! That's a recipe for disaster (and maybe a ruined cake, speaking from experience).

The Vanilla Extract Dilemma (Resolved!)
Back to my baking adventure! I did some frantic Googling and eventually found the density of my specific brand of vanilla extract. It turned out to be something like 0.97 grams per milliliter. (Remember to find your specific brand – they vary!).
Converting that to micrograms per milliliter (0.97 g/mL * 1,000,000 µg/g = 970,000 µg/mL), I could finally calculate how many milliliters of vanilla extract I needed to get my 300 micrograms.
Mass = Density * Volume
Volume = Mass / Density

Volume = 300 µg / 970,000 µg/mL
Volume ≈ 0.000309 mL
Turns out, it was a TINY amount. Like, a ridiculously tiny amount. Good thing I had a pipette! The cake, by the way, was... well, let's just say it was edible. (Next time, I'm simplifying the recipe.)
Key Takeaway: You cannot directly convert milliliters to micrograms without knowing the density of the substance. Find the density, do the math, and happy measuring! (And maybe stick to simpler recipes. Just a thought.)
