Co3 2 Resonance Structures

Alright, settle in, chemistry newbies and seasoned science enthusiasts alike! Today, we're diving into a topic that might sound intimidating but is actually quite chill: the resonance structures of CO32-, aka the carbonate ion. Think of it as the Beyoncé of polyatomic ions – iconic, powerful, and with a few different looks that all contribute to its overall fabulousness.
What's the Buzz About Resonance?
So, what is resonance anyway? Imagine you're trying to describe your best friend's style. You could say they're "bohemian," or "chic," or "edgy." Each describes a different facet, but none perfectly captures the whole vibe. Resonance is similar. It's when a single Lewis structure just can't accurately depict the bonding in a molecule or ion. Instead, we use multiple Lewis structures, called resonance structures, to represent the true electron distribution.
These aren't different versions of the molecule popping into existence. Instead, think of the actual molecule as a hybrid, a kind of blend or average of all the resonance structures. It's like looking at a blended smoothie – you can see hints of the banana, the berries, and the spinach, but it's all one delicious, integrated concoction.
Must Read
CO32-: A Carbonate Case Study
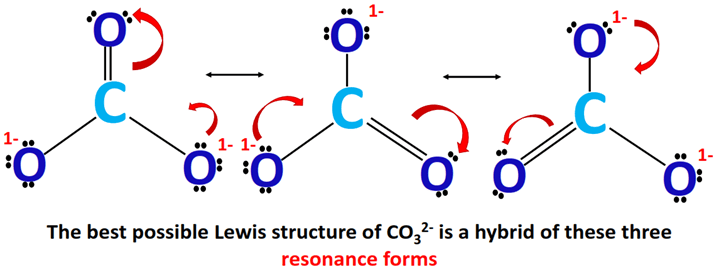
Let's bring this back to our star of the show, the carbonate ion (CO32-). Carbonate has one carbon atom bonded to three oxygen atoms. Here's where it gets interesting: to satisfy the octet rule (the chemical law that atoms "want" eight valence electrons), one of the carbon-oxygen bonds has to be a double bond, while the other two are single bonds. But which oxygen gets the double bond?
That's where resonance comes in. Instead of picking one oxygen to hog the double bond, we draw three possible Lewis structures. In each structure, the double bond is on a different oxygen. These are our resonance structures. We connect them with a double-headed arrow (↔) to show that they're resonance forms, not different molecules.

Pro Tip: Remember that the atoms themselves don't move! Only the electrons do. It's like switching the lead guitarist in a band – the rest of the band stays put, but the sound changes slightly.
Why This Matters: The Real World Perks
Okay, so we've drawn some Lewis structures. Big deal, right? Actually, it is a big deal. Understanding resonance helps us understand why carbonate ions are so stable and how they behave. Here's the breakdown:

- Bond Lengths: In reality, all three carbon-oxygen bonds in carbonate are the same length. This wouldn't be true if one was a true double bond and the others were single bonds. Resonance explains this by telling us each bond is partially double-bonded, somewhere between a single and a double bond.
- Stability: Resonance stabilizes molecules. The more resonance structures, the more stable the molecule generally is. This stability is important in many chemical reactions. Think of it like having a strong foundation for a building – the more solid the foundation, the less likely it is to crumble.
- Everywhere Around Us: Carbonate ions are found in everything from limestone and chalk to baking soda and antacids! They're essential components in the Earth's crust, play a crucial role in regulating pH levels, and are involved in various biological processes.
Resonance in Pop Culture (Kind Of)
Okay, this is a stretch, but bear with me. Think of the multiverse in movies like "Spider-Man: Into the Spider-Verse" or "Everything Everywhere All at Once." Each universe is a different possibility, a different way things could be. Similarly, each resonance structure is a different possibility for how the electrons could be arranged. The actual molecule is like the character hopping between universes, experiencing a blend of all the possibilities. See? Chemistry is entertainment!
Fun Fact: The concept of resonance was first introduced by Linus Pauling, a Nobel laureate known for his work on chemical bonding. He was a real rockstar of the science world!
A Little Reflection
So, what's the takeaway from our journey into the world of carbonate resonance? It's a reminder that things aren't always as simple as they seem. Sometimes, a single perspective isn't enough to capture the whole truth. Whether you're thinking about chemical bonds or human relationships, it often takes multiple viewpoints and a bit of flexibility to truly understand the bigger picture. Embracing different perspectives, just like embracing all resonance structures, can lead to a more complete and accurate understanding of the world around us. And who knows, maybe it'll even help you ace your next chemistry quiz.