Intermolecular Forces Are Primarily Responsible For:

Hey there, science buddy! Ever wonder why water beads up on a freshly waxed car? Or why you can actually walk on water (well, tiny insects can, anyway)? The answer, my friend, lies in the mysterious world of intermolecular forces (IMFs)! Don't run away screaming just yet – I promise it's way more interesting than it sounds. We're not talking about some kind of intergalactic war, just the tiny forces holding molecules together.
So, what are these IMFs primarily responsible for, you ask? Let's dive in! Think of them as the secret agents behind the scenes, dictating how substances behave in their everyday lives.
Boiling Points and Melting Points: Hot Stuff!
First up, and arguably most importantly, IMFs are a major player in determining a substance's boiling point and melting point. Imagine trying to separate two magnets stuck together. The stronger the magnets, the more effort (energy) you need to pull them apart, right? It's the same principle with molecules! Stronger IMFs mean it takes more heat (energy) to break those attractions and make a substance melt or boil.
Must Read
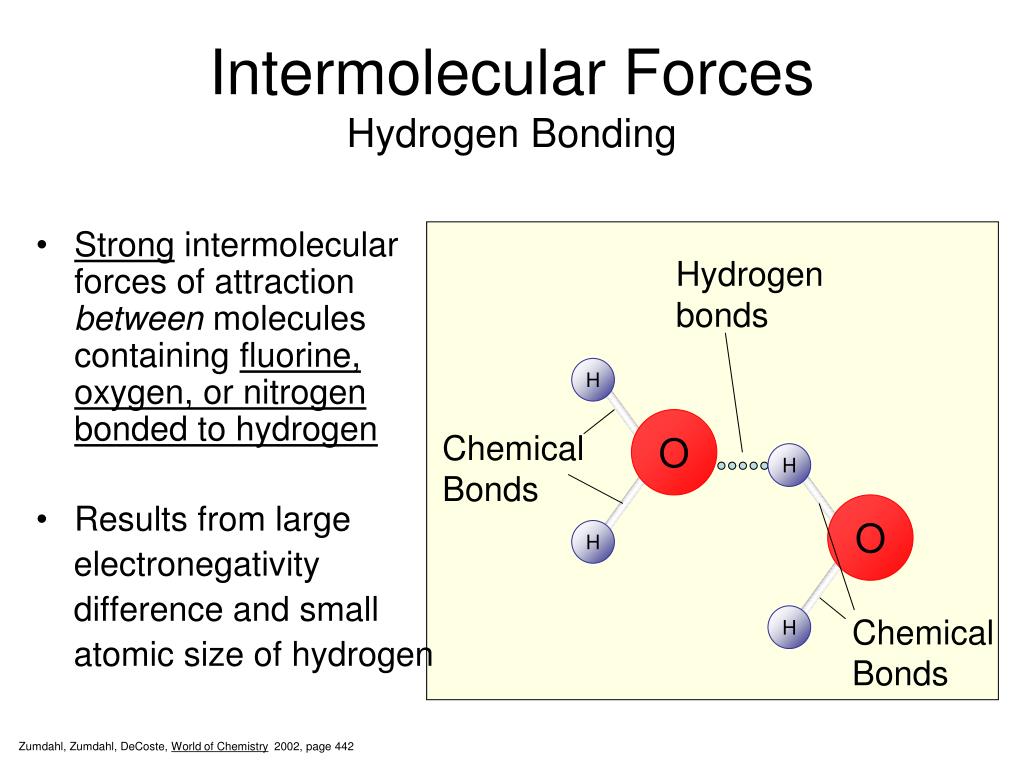
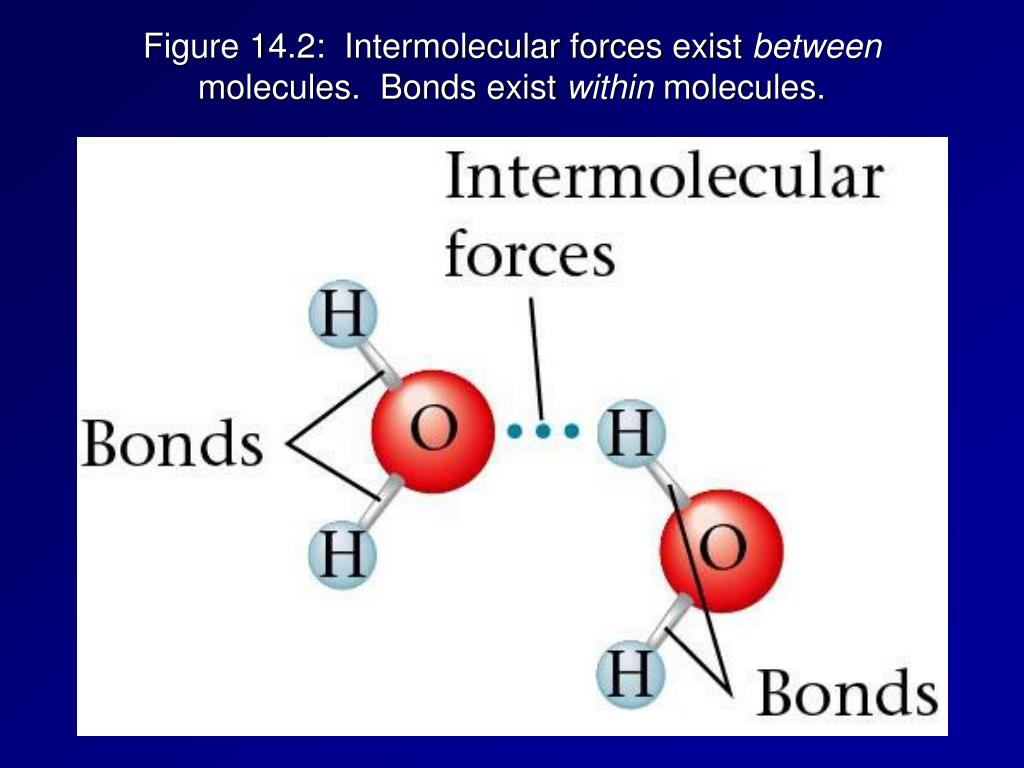
That's why water (with its relatively strong hydrogen bonding – a type of IMF) has a much higher boiling point than, say, methane (a gas at room temperature with very weak IMFs). Water molecules are holding on tight! Methane is all like, "Peace out! I'm outta here at -161°C!".
Seriously though, if it weren't for hydrogen bonds, water would be a gas at room temperature. Kiss goodbye to swimming pools, and probably life as we know it! (Dramatic, I know, but true!).

States of Matter: Solid, Liquid, or Gas?
Linked closely to boiling and melting points, IMFs dictate whether a substance is a solid, liquid, or gas at a given temperature. Solids have the strongest IMFs, holding the molecules tightly in a fixed arrangement. Liquids have weaker IMFs, allowing molecules to move around more freely, but still stick together to some extent. Gases have the weakest IMFs, allowing molecules to zoom around independently, with very little attraction to each other. Think of it like a dance floor: solids are all slow dancing close together, liquids are doing the cha-cha with a little more space, and gases are moshing in a free-for-all.
So, if you're wondering why iron is solid and oxygen is a gas (at room temperature, of course), thank the IMFs!
Surface Tension: Walking on Water (Sort Of)
Remember that water beading up? That's surface tension in action! It's like a tiny film on the surface of the water, created by the cohesive forces (IMFs) between water molecules. These forces pull the surface molecules inwards, minimizing the surface area and creating that beading effect. This is why some insects can skitter across the surface of water – they're light enough that the surface tension can support their weight.
Think of it like a bunch of friends holding hands really tightly – they can support a lightweight person walking across their arms! Now, I wouldn’t recommend trying it yourself in your local swimming pool!

Viscosity: How Thick is Too Thick?
Ever tried pouring honey? It's…slow. That's because honey has a high viscosity. Viscosity is a measure of a fluid's resistance to flow, and it's heavily influenced by IMFs. The stronger the IMFs, the more difficult it is for molecules to slide past each other, resulting in a higher viscosity.
Water has relatively low viscosity because its IMFs are weaker than honey's. Try comparing pouring water to pouring molasses – the difference is stark! It's the same reason why motor oil comes in different viscosities; depending on your engine and the climate, you need oil that flows (or doesn't flow) at a certain rate.
Solubility: Mix It Up!
IMFs also play a critical role in solubility – whether one substance will dissolve in another. The general rule of thumb is "like dissolves like." This means that substances with similar IMFs tend to be soluble in each other. For example, water (a polar molecule with hydrogen bonding) dissolves polar substances like sugar. Oil (a nonpolar molecule) doesn't dissolve in water because their IMFs are too different. However, oil dissolves other nonpolar substances like grease. Think of it as molecules wanting to hang out with their own kind – birds of a feather, and all that!
So there you have it! Intermolecular forces: the unsung heroes of the molecular world, quietly dictating the properties of the stuff around us. They are the reason why things are the way they are.
Understanding these forces helps us understand the world better. So the next time you see water beading on your car, remember those tiny little IMFs working their magic behind the scenes. They might be small, but they sure are powerful! Aren't you glad you know more about them now?
