Ammonium Nitrate Molar Mass

Alright, gather 'round, folks! Let me tell you a tale. A tale of…ammonium nitrate! Now, I know what you’re thinking: "Nitrogen fertilizer? Sounds thrilling as watching paint dry!" But hold your horses, because we're diving into the nitty-gritty of its molar mass, and trust me, it’s way more interesting than it sounds – especially when I’m telling the story.
So, what exactly is molar mass? Well, imagine you're baking a cake, but instead of cups and tablespoons, you're using…moles! No, not the furry little diggers in your backyard, but the chemical kind. A mole is just a fancy way to say a huge number of molecules (specifically, 6.022 x 10^23, also known as Avogadro's number). It's like a baker's dozen, but on steroids. Think of it as the ultimate party pack of molecules.
And molar mass? That's just the weight of one mole of a substance. Easy peasy, right? It’s measured in grams per mole (g/mol). Basically, how much a whole party pack of a particular molecule weighs.
Must Read
Ammonium Nitrate: The Star of Our Show
Now, let's zoom in on our star: ammonium nitrate (NH₄NO₃). Sounds like something out of a sci-fi movie, doesn't it? Don’t worry, it's not going to morph into a giant robot anytime soon (probably). But it's got some serious chemical mojo.
First, we need to break this down, like dismantling a complicated IKEA bookshelf (without the instructions, obviously!). We need to see what elements make up ammonium nitrate:
- Nitrogen (N): We've got two of these bad boys! Nitrogen is what makes our atmosphere breathable... mostly.
- Hydrogen (H): Four of these little guys! Hydrogen is the lightest element, and a real lightweight in this calculation, too.
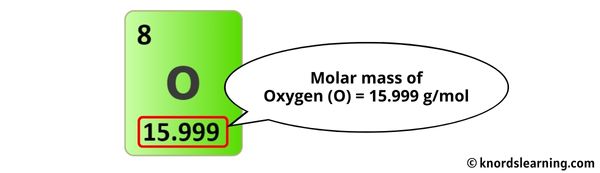
- Oxygen (O): Three oxygen atoms! Oxygen is what keeps us alive, and is also responsible for fire. Bit of a Jekyll and Hyde situation, really.
The Atomic Weight Detective Work
Okay, so we know what we have, but now we need to know how much each atom weighs. This is where the periodic table comes in. Think of it as the ultimate cheat sheet for chemists. It's got all the atomic weights listed nice and neatly. Just find your element and bam! You've got your weight.
Here's the atomic weight lineup (rounding to two decimal places for simplicity because ain’t nobody got time for super-precise calculations during story time):
- Nitrogen (N): 14.01 g/mol
- Hydrogen (H): 1.01 g/mol
- Oxygen (O): 16.00 g/mol
Remember, these are the weights of one mole of each atom. So, one mole of nitrogen atoms weighs about 14.01 grams. Make sense?
The Grand Calculation: Prepare for Numbers!
Now for the moment of truth! Time to crunch those numbers. Remember our formula, NH₄NO₃? We’re going to multiply the atomic weight of each element by the number of times it appears in the formula, and then add it all up! Imagine it like calculating the total weight of your luggage at the airport – except your luggage is made of atoms.
Here we go:
- Nitrogen: 2 atoms * 14.01 g/mol = 28.02 g/mol
- Hydrogen: 4 atoms * 1.01 g/mol = 4.04 g/mol
- Oxygen: 3 atoms * 16.00 g/mol = 48.00 g/mol
Now, add 'em all together:

28.02 g/mol + 4.04 g/mol + 48.00 g/mol = 80.06 g/mol
Ta-da! The molar mass of ammonium nitrate is approximately 80.06 g/mol. That means one mole (that incredibly huge party pack) of ammonium nitrate weighs about 80.06 grams.
Why Should I Care About This Molar Mass Stuff?
Okay, you might be thinking, "So what? Why did I just spend the last few minutes learning about this?" Well, knowing the molar mass is super important for…well, chemists! They use it to figure out how much of a substance they need for reactions. Think of it as a crucial ingredient in the recipe for creating all sorts of cool (and sometimes not-so-cool) things.
Also, it’s a great conversation starter at parties. Just imagine: “Hey, did you know the molar mass of ammonium nitrate is 80.06 grams per mole?” You’ll be the hit of the night…or maybe not. But you’ll definitely have some interesting facts to share.
So, there you have it! The thrilling (hopefully!) tale of the ammonium nitrate molar mass. Now you can impress your friends, family, and even your pet hamster with your newfound chemical knowledge. You're basically a chemistry genius now. Go forth and calculate!
And remember, chemistry is like cooking, but don't lick the spoon... unless you really know what you're doing.
