How Many Energy Levels Are There
Okay, picture this: you're at the biggest, most epic concert EVER! Imagine a stadium that stretches beyond the horizon, filled with more people than you thought existed. This is kinda like an atom, but instead of people, we have electrons!
So, What Are Energy Levels?
Think of energy levels like VIP sections at our mega-concert! Some sections are closer to the stage (more energy), and others are farther away (less energy). The closer you are, the better the view, the more pumped you are – that's high energy!
Electrons, being the concert-loving particles they are, prefer to hang out in specific spots. These are our energy levels, also known as electron shells or orbitals. It's like assigned seating, but way cooler.
Must Read
Imagine throwing a bouncy ball at a set of stairs. The ball won't stop in between steps, right? It lands squarely on a step. Electrons are kinda the same way. They don't just hang out between energy levels; they occupy specific, defined spots!
The Principal Quantum Number: Your VIP Pass
Each energy level has a number, cleverly called the principal quantum number, often represented by the letter 'n'. It’s basically the number on your VIP pass! n=1 is the closest to the nucleus, then n=2, n=3, and so on. The higher the 'n', the further away from the nucleus, and the higher the energy!
Think of it like a multi-story house! The ground floor (n=1) is the easiest to reach, but the penthouse suite (n=100... well, theoretically!) has the best view and costs a fortune in energy.
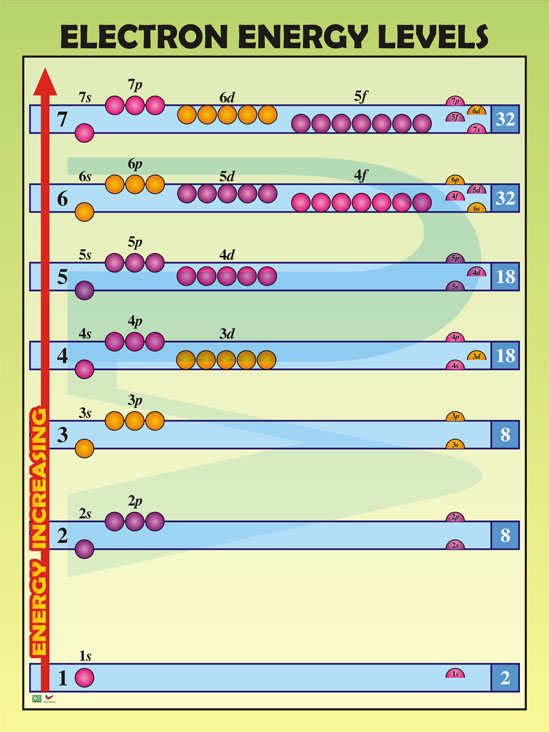
How Many Energy Levels ARE There, Anyway?
This is the million-dollar question! Buckle up, because the answer is... practically infinite, but realistically, it depends.
In theory, an atom could have an infinite number of energy levels extending outwards. Imagine a stadium expanding forever! But let’s get practical.

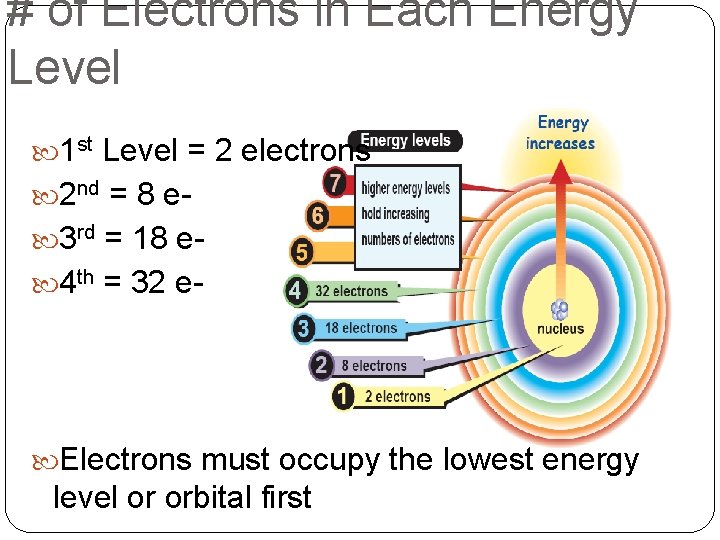
The number of energy levels an atom actually uses depends on how many electrons it has. An atom will only fill up the energy levels that have electrons in them. You can't have a party on the tenth floor of your building if you don't have enough guests to fill even the first floor!
Even with the heaviest elements we know (like those super-unstable ones created in labs), we don't need to invoke levels past n=7. That's right, level seven. That super high level is for only the most dedicated of electrons.
The Limit Does Exist (Sort Of)
So, practically speaking, we're usually dealing with a maximum of 7 energy levels. This is because no element found (so far!) requires more than 7 to hold all its electrons. We are all practical, in our own unique way.
However, that's not to say that higher energy levels don't exist! They absolutely do. It's like having extra rooms in your house that you only use for special occasions (or when your in-laws visit). They're there, just not always occupied.
Imagine an electron jumping from level 1 (close to the nucleus) to level 7 (way out there!). This requires a HUGE amount of energy, like winning the lottery and using it to buy a private island. Possible, but not exactly common.
Sublevels: The Inner Circle
Hold on, there's a VIP section within each VIP section! These are called sublevels, and they're even more exclusive. Think of them like having different types of seats in the stadium, like regular seats, box seats, and front-row access.
Each energy level (n) has 'n' number of sublevels. So, level 1 (n=1) has one sublevel, level 2 (n=2) has two sublevels, level 3 (n=3) has three, and so on.
These sublevels are named using letters: s, p, d, and f. They each have different shapes and hold different numbers of electrons. It’s like having different types of party rooms within the stadium, each with its own vibe!
The s sublevel is like a small, cozy room that can hold a maximum of 2 electrons (perfect for a close couple!). The p sublevel is a bit larger and can hold 6 electrons (a small group of friends). The d sublevel holds 10 electrons (a lively party), and the f sublevel holds a whopping 14 electrons (a full-blown rave!).
You don’t need to remember these numbers necessarily. But, you do know that the more energy that is involved, the bigger the party gets.

Orbital Overload!
Each sublevel is further divided into orbitals. These are like individual chairs within each party room. Each orbital can hold a maximum of two electrons, but they must have opposite "spins" (think of them as spinning in opposite directions to avoid bumping into each other).
So, an s sublevel has 1 orbital, a p sublevel has 3 orbitals, a d sublevel has 5 orbitals, and an f sublevel has 7 orbitals. It’s like organizing seating arrangements for your ridiculously large electron party!
Why Does This Matter?
Understanding energy levels, sublevels, and orbitals is crucial for understanding how atoms interact with each other. It's the foundation of chemistry!
The way electrons are arranged determines how an atom will bond with other atoms to form molecules. It dictates whether a substance is reactive, stable, or something in between. It's like understanding the personalities of the guests at your party to make sure everyone gets along!
From the simplest reactions (like burning a piece of paper) to the most complex processes (like photosynthesis in plants), energy levels play a fundamental role. It’s all about the electrons and their dance around the nucleus.

So, next time you see a fireworks display or cook a meal, remember the incredible dance of electrons happening at the atomic level. It’s a constant exchange of energy, governed by the rules of those magical energy levels.
In Conclusion: A Celebration of Energy Levels
While the number of energy levels can theoretically be infinite, we generally deal with a maximum of 7 in real-world elements. These levels, along with their sublevels and orbitals, dictate the behavior of atoms and molecules, shaping the world around us.
Think of each atom as a tiny, self-contained universe, with electrons orbiting the nucleus like planets around a star. It's a beautiful and complex system, and understanding energy levels is key to unlocking its secrets.
So go forth and explore the amazing world of atomic structure! And remember, whether you're at the closest VIP section or somewhere further back, the energy of the universe is with you.
