Write The Complete Ground-state Electron Configuration Of Aluminum.
Hey there, science enthusiast! Ever wonder what atoms are really doing inside your phone, your desk, or even you? Let's dive into something super fundamental: electron configurations! Specifically, let's tackle aluminum. Yes, that silvery metal making up your soda can (probably!).
What's the Big Deal with Electron Configurations?
Think of electron configurations as the atomic equivalent of a seating chart. Except, instead of students, we're organizing tiny, negatively charged particles called electrons. These electrons zip around the atom's nucleus in specific energy levels and orbitals. It’s like an electron rave in a super tiny club!
Knowing how these electrons are arranged is a big deal! It dictates how an atom will interact with other atoms, forming bonds, creating molecules, and generally making the world (and your soda can) possible. Chemistry, baby! It’s all about the electron dance.
Must Read
Aluminum: Our Star Today
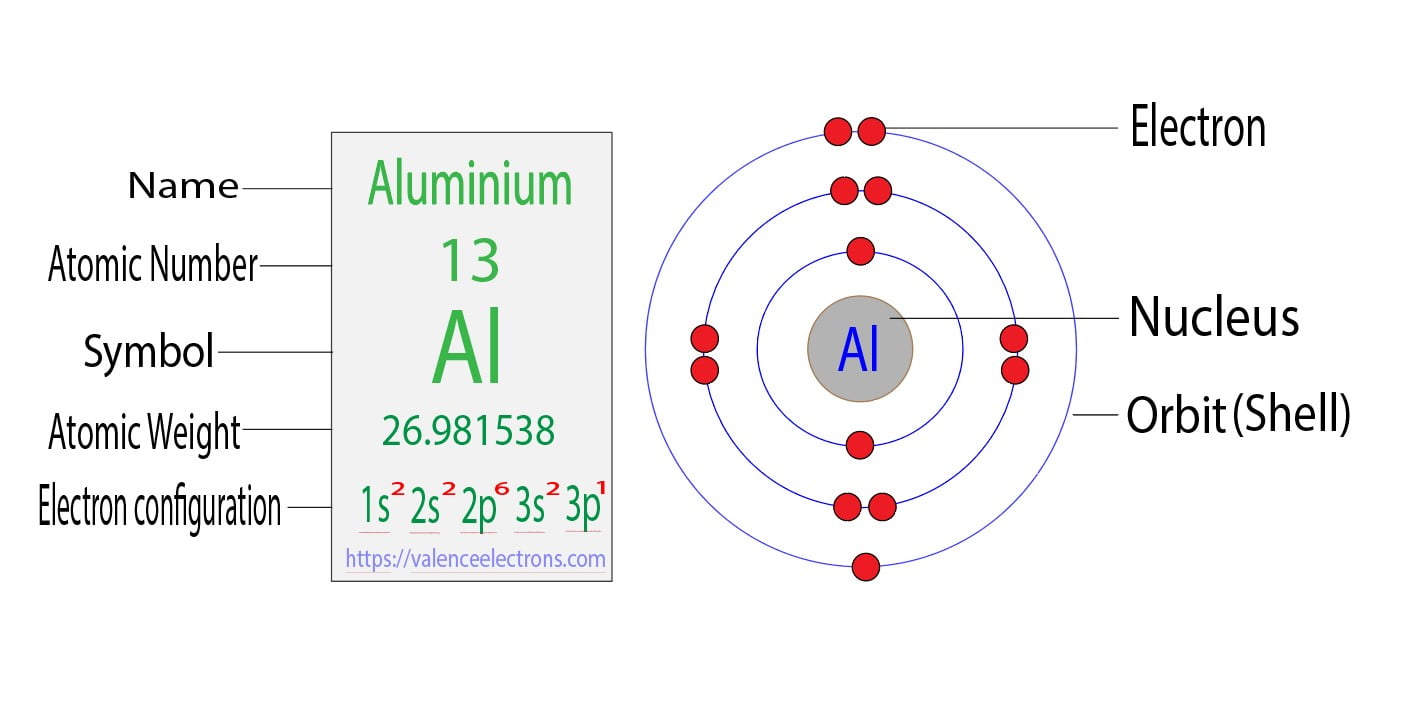
Aluminum (Al) is element number 13. This crucial piece of info means it has 13 protons in its nucleus. Because atoms like to be neutral (usually!), it also has 13 electrons buzzing around. That's our target: figuring out where those 13 electrons are chilling.
Quick aside: Aluminum is everywhere. From foil to airplane parts, it's a lightweight, strong, and versatile metal. Plus, recycling it is super energy-efficient! High five, aluminum!
The Rules of the Electron Game
Okay, so there are a few rules to this electron seating chart: * Electrons fill the lowest energy levels first. It's like wanting the best seat in the house! * Each orbital (a specific region of space) can hold a maximum of two electrons. Think of it as a cozy, electron-duo zone. * There are different types of orbitals: s, p, d, and f. Each has a different shape and energy. S orbitals are spherical, p orbitals are dumbbell-shaped, and d and f...well, they get complicated! Let’s not go there right now.
Think of it like musical chairs, but with electrons and predetermined spots. May the best electron win!...by landing in the lowest energy chair.
Building the Aluminum Configuration, Step-by-Step
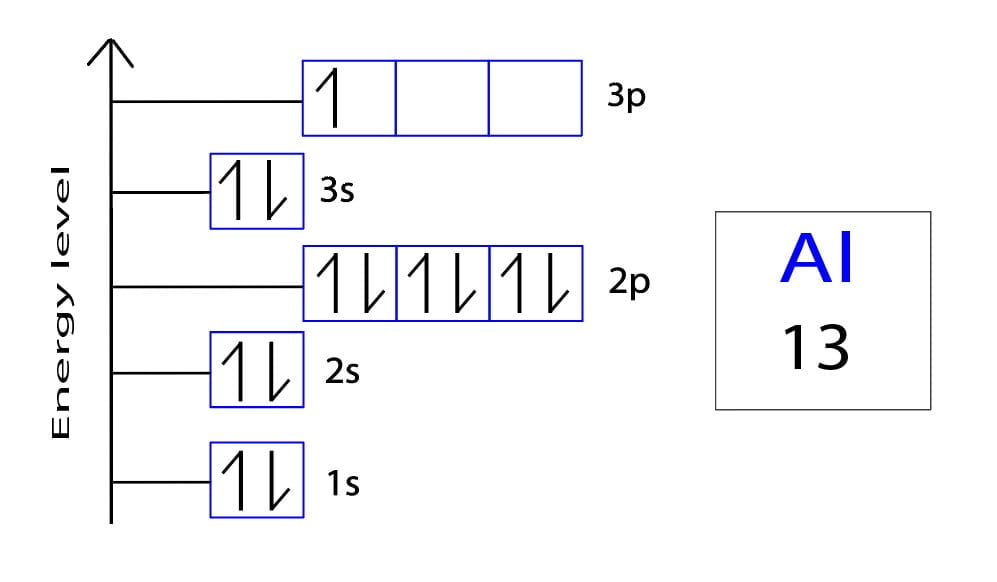
Let's put those 13 electrons into their spots, level by level: * 1s: This is the lowest energy level. It has only one s orbital, which can hold 2 electrons. So, we fill it up: 1s2. Two electrons down, 11 to go! * 2s: Next up is the 2s orbital, also holding a max of 2 electrons: 2s2. Now we have 1s22s2. Four electrons chilling, 9 still need placement. * 2p: Here's where things get a little spicier! The 2p level has three p orbitals. Each orbital holds 2 electrons, so the 2p sublevel can hold a total of 6 electrons. Let’s fill ‘er up! 2p6. We're at 1s22s22p6. We've seated 10 electrons. Only three left! * 3s: Back to an s orbital! This 3s orbital holds 2 electrons: 3s2. Our configuration is now 1s22s22p63s2. We're down to just one electron left to place! * 3p: Finally, we reach the 3p sublevel. Remember, it can hold six, but we only have one electron left. So, it gets its own private room! 3p1.

And there you have it! The complete ground-state electron configuration of aluminum is:
1s22s22p63s23p1
That's the final answer! This is the most stable, lowest-energy arrangement for aluminum's electrons.
Shortcuts and Other Fun Facts
There's a shortcut notation using noble gases! Since 1s22s22p6 is the same as the electron configuration of neon (Ne), we can write aluminum's configuration as [Ne]3s23p1. Pretty cool, huh?
The electrons in the outermost energy level (in aluminum's case, the 3s23p1 electrons) are called valence electrons. They're the most important for chemical bonding. Aluminum has 3 valence electrons, which explains why it readily forms bonds with other elements.
Extra credit: The term "ground state" simply means the lowest energy state. Atoms can get "excited" by absorbing energy, causing electrons to jump to higher energy levels. But they eventually fall back down to the ground state, releasing energy (often as light!). Think fireworks!
Why Should You Care?
Understanding electron configurations helps you understand… well, almost everything! From why certain elements are shiny to how medicines work, it all boils down to how electrons are arranged. It's the fundamental language of chemistry.
So, the next time you crack open a can of soda, take a moment to appreciate the 13 electrons diligently orbiting the nucleus of each aluminum atom. They're doing their part to keep your drink cold (and your brain buzzing with scientific curiosity!).
