Why Does Splitting An Atom Release Energy
Ever wondered why splitting an atom is such a big deal? Like, seriously, why does it cause such a massive release of energy? It's not just some science fiction mumbo-jumbo; it's real, it's powerful, and understanding it can seriously blow your mind (in a good way, of course!).
So, let's dive in, shall we? No need for a Ph.D. in physics here; we'll keep it simple and fun. Forget those complicated equations you might remember (or tried to forget!) from high school. We're talking about the basics, the really cool stuff that makes you go, "Aha!".
The Mighty Atom: It's All About the Nucleus
At the heart of every atom is the nucleus. Think of it as the atom's tiny, super-dense control center. Inside this nucleus, you'll find protons and neutrons, which are collectively known as nucleons. These little guys are held together by the strong nuclear force, an incredibly powerful force that, well, strongly attracts them to each other.
Must Read
Now, here’s where things get interesting. Imagine you’re trying to break apart a really strong magnet. You'd have to use a lot of energy, right? The same principle applies to the nucleus. It takes energy to pull those protons and neutrons apart.
But wait! If it takes energy to pull them apart, why does splitting the atom release energy? This is the million-dollar question (or, perhaps, the multi-trillion-dollar question, considering the power of nuclear energy!).

Mass-Energy Equivalence: Einstein to the Rescue!
Enter Albert Einstein and his famous equation: E = mc². You've probably seen it before, maybe on a t-shirt or in a movie. But what does it actually mean? Simply put, it means that energy (E) is equivalent to mass (m) multiplied by the speed of light (c) squared.
The speed of light is, well, incredibly fast. So, even a tiny amount of mass can be converted into a huge amount of energy. Think about it: a tiny pebble might not seem like much, but if you could convert its mass entirely into energy, you could power a city!
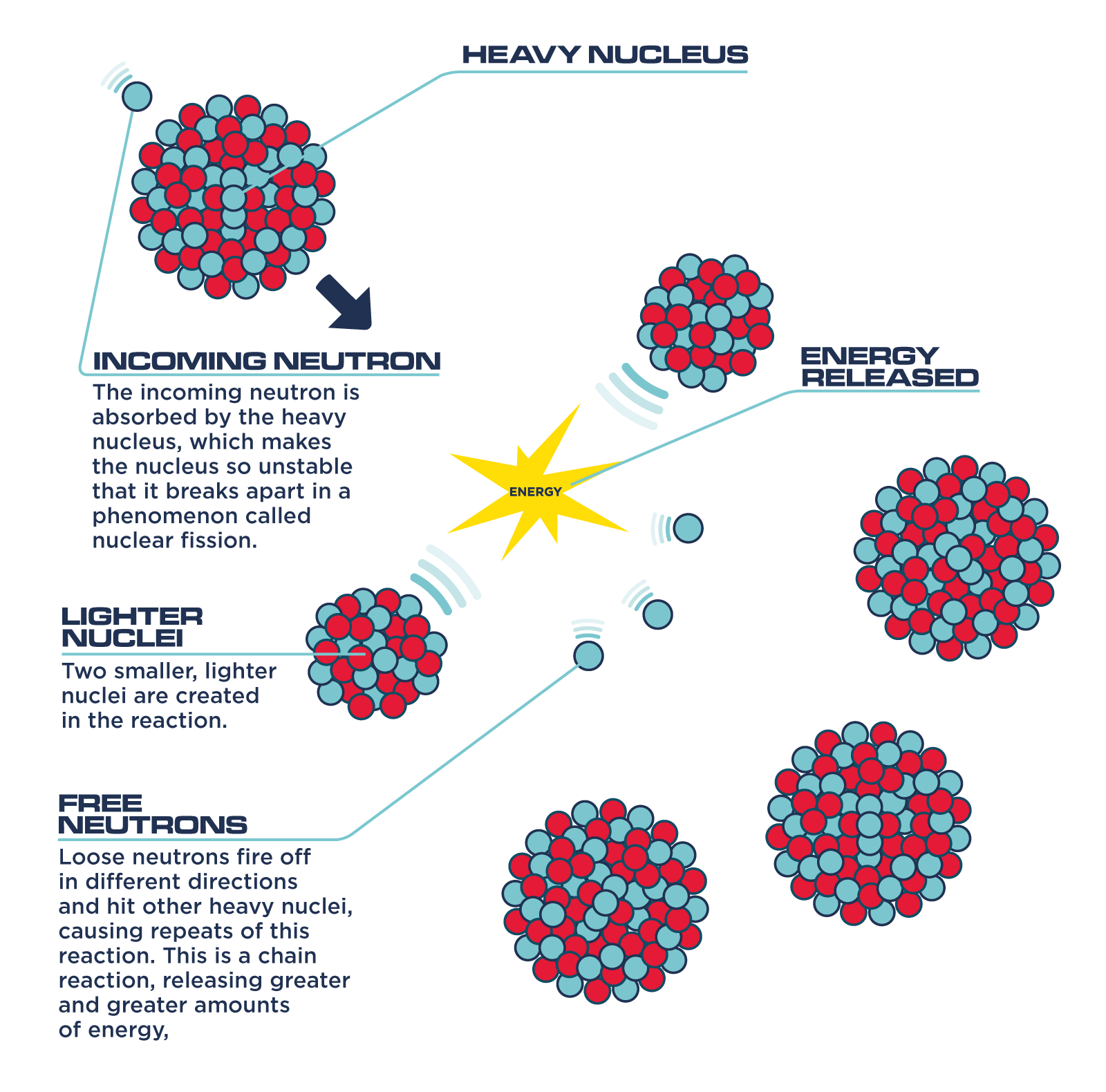
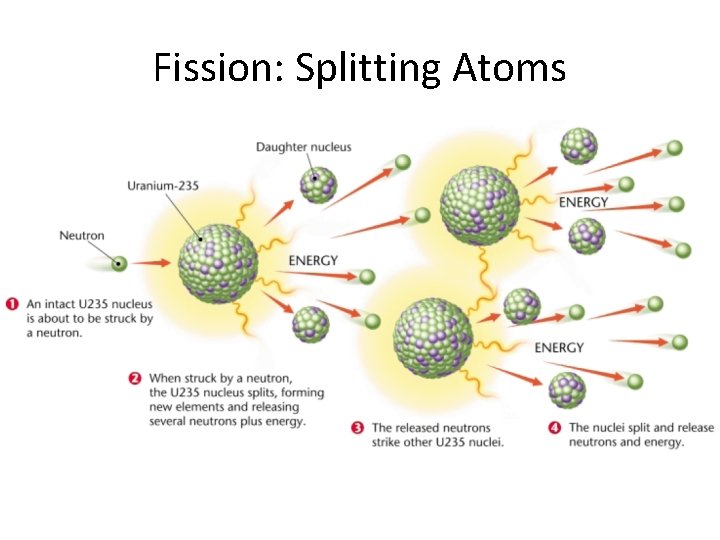
So, back to splitting the atom. When you split a heavy atom like uranium, the resulting pieces (smaller atoms and some extra neutrons) actually have slightly less mass than the original uranium atom. Where did that mass go? You guessed it: it was converted into energy!
Binding Energy: The Key to Unlocking the Power
Another crucial concept is binding energy. This is the energy that holds the nucleus together. Different atoms have different binding energies per nucleon. Iron, for example, has a very high binding energy per nucleon, meaning it's very stable.
When you split a heavy atom, the resulting smaller atoms have a higher binding energy per nucleon than the original heavy atom. This means they are more stable. The difference in binding energy is released as kinetic energy – that is, the energy of motion – of the resulting fragments and neutrons, and also as gamma rays (a form of electromagnetic radiation).

Think of it like this: imagine you have a bunch of LEGO bricks loosely connected. They have a certain amount of "binding energy" holding them together. Now, imagine you rearrange them into a more stable, tightly packed structure. The rearranged structure has a higher binding energy. The excess energy is released – maybe as a little click as the pieces snap together more securely.
Chain Reactions: Unleashing the Beast
But wait, there's more! The neutrons released when you split an atom can then go on to split other atoms, leading to a chain reaction. This is what happens in nuclear reactors and, unfortunately, also in atomic bombs. It's a self-sustaining process that releases a tremendous amount of energy.
So, splitting the atom isn't just about breaking something apart. It's about converting a tiny amount of mass into a staggering amount of energy, thanks to Einstein's genius and the amazing properties of the atomic nucleus.

Isn't that amazing? It might seem complicated at first, but once you grasp the basic principles, it's incredibly fascinating. And understanding these concepts can help you appreciate the world around you in a whole new way.
Beyond the Basics: Fueling Our Curiosity
Now, I know what you're thinking: "This is cool, but I want to know more!". And that's fantastic! The world of nuclear physics is vast and exciting. There's so much more to explore, from nuclear fusion (the process that powers the sun) to the development of new nuclear technologies for medicine and energy.
So, go out there, explore, and never stop learning! Who knows? Maybe you'll be the one to discover the next groundbreaking innovation in nuclear science. The possibilities are truly endless!
