Why Does An Ion Have A Charge

Hey there, fellow curious human! Ever wonder what gives that amazing zing to your favorite lemon water, or why your hair occasionally decides to stage a gravity-defying revolt after a particularly enthusiastic sweater removal? What about the hidden power in your phone battery, or the very essence of why salt tastes, well, salty?
The answer, my friends, is all thanks to something called an ion. And trust me, once you unravel this little mystery, the world around you suddenly starts to click in a whole new, utterly fascinating way!
The Tiny World of Atoms: Where It All Begins
Let's start with the basics, shall we? Everything around us, including you, me, and that perfectly brewed cup of coffee, is made up of incredibly tiny building blocks called atoms. Now, these aren't just solid little marbles; oh no, they're bustling, mini-universes themselves!
Must Read
At the very heart of an atom, you'll find the nucleus, packed with two types of particles: the positively charged protons (think of them as the life of the atomic party, always bringing the positive vibes) and the neutral neutrons (the calm, chill buddies who just hang out). Orbiting around this nucleus at lightning speed are the tiny, super energetic electrons – and these little rebels are negatively charged.
Got that? Protons are positive, electrons are negative, and neutrons are, well, neutral. Simple enough, right?
The Art of Balance: Why Most Atoms Are Neutral
Normally, in a happy, healthy, and perfectly balanced atom, the number of positive protons perfectly matches the number of negative electrons. It's like a perfectly choreographed dance, or a cosmic tug-of-war where neither side wins. The positive charges cancel out the negative charges, resulting in a perfectly neutral atom. No charge, no fuss, just harmonious existence.

These neutral atoms are super content, just chilling in their uncharged state. They're like the quiet observer at a party, perfectly happy to just be there without drawing attention.
Enter the Ion: When Atoms Get Charged Up!
But what if that perfect balance gets disrupted? What if an atom decides it's time for a little change, a little shake-up? This, my dear reader, is where the incredible magic of ions happens! An ion is simply an atom (or even a group of atoms joined together) that has either gained or lost one or more electrons.
"But why would they do that?" you ask, and it's an excellent question! Atoms, like all of us, are striving for stability. They want to achieve a "full house" in their outermost electron shell, a state of perfect contentment. Sometimes, it's easier to shed a few electrons to get there, and other times, it's simpler to invite a few more in.

The Two Types of Charged Superstars: Cations and Anions
When an atom decides to get rid of one or more of its negatively charged electrons, it's suddenly left with more positive protons than negative electrons. Think about it: if you lose a negative thing, you become more positive overall, right? The result? A net positive charge!
We call these positive pals cations. They're like the generous souls who shed their baggage and feel lighter and more positive for it. A great example is sodium (Na), which often loses an electron to become a sodium ion (Na⁺).
On the flip side, what if an atom decides to gain one or more electrons? Now it has more negative electrons than positive protons. Voila! A net negative charge! These negatively charged characters are known as anions.
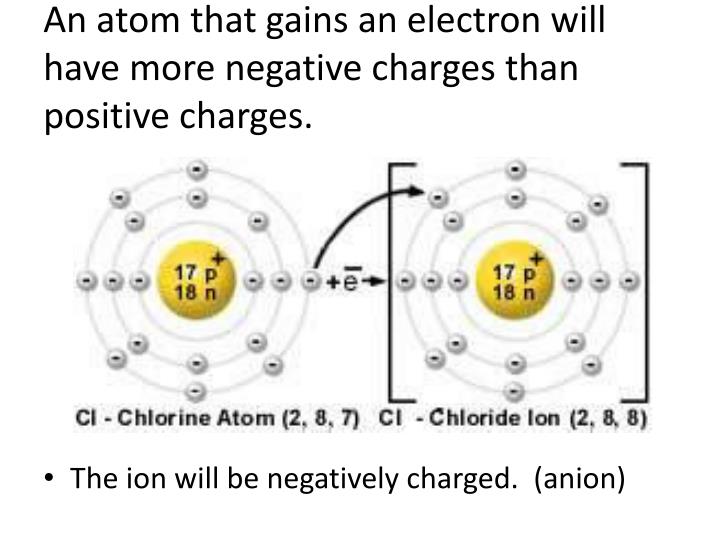
They're like the meticulous collectors, gathering more to their inner circle. Chlorine (Cl), for instance, loves to gain an electron to become a chloride ion (Cl⁻).
And here’s the really, truly important bit, a little secret just between us: during these electron-swapping transformations, the number of protons never changes. If it did, you wouldn't just have a charged atom; you'd have an entirely different element! It's always about the electrons playing musical chairs.
Ions in Action: Making Your Life More Fun!
So, why should you, a wonderfully curious individual, care about these tiny, charged rebels? Because they're absolutely everywhere and they're doing everything! Understanding them isn't just for scientists in lab coats; it's for you. It's a superpower that lets you see the hidden chemistry in your everyday life.

- Electrolytes: That sports drink you gulp after a workout? It's packed with ions like sodium (Na⁺), potassium (K⁺), and chloride (Cl⁻)! These aren't just fancy words; they're essential for nerve function, muscle contraction, and keeping you hydrated. Suddenly, your thirst quencher is a scientific marvel!
- Salty Snacks: The very reason salt (sodium chloride) tastes salty? It's because when it hits your tongue, it breaks apart into Na⁺ and Cl⁻ ions, which your taste buds detect. Mind. Blown.
- Batteries: Yep, your phone, your laptop, even your car battery – they all rely on the precise, controlled movement of ions to generate electrical current and power your world. It's an ion highway!
- Static Cling: Remember rubbing that balloon on your hair and getting a wild hair day? You were literally transferring electrons between the balloon and your hair, creating static ions and causing those hilarious repulsive forces!
- Your Body! Believe it or not, your very existence is powered by ions. Nerve impulses, muscle contractions, the balance of fluids in your cells – it’s all an intricate dance of charged particles making sure you can think, move, and even read this article!
Embrace the Wonder, Charge Up Your Curiosity!
See? Understanding why an ion has a charge isn't just some dusty textbook fact. It's a key that unlocks a deeper appreciation for the world around us. It transforms the mundane into the magnificent, turning everyday occurrences into exciting demonstrations of fundamental physics and chemistry.
So, the next time you taste something salty, feel a spark of static electricity, or simply gulp down a refreshing drink, give a little nod to the humble ion. It’s a tiny player on a grand stage, showing us that even the smallest imbalance can lead to astonishing power, beauty, and the very fabric of existence.
Go forth and marvel! Let your curiosity be charged, and never stop asking "why?" The universe is waiting to reveal its incredible secrets, and you're already one step closer to understanding its electrifying dance.
