Why Are Interstitial Alloys Less Malleable

Ever wondered why that awesome, super-strong steel bridge doesn't just bend and buckle in the wind like a flimsy paperclip? Or why the sturdy frame of your bicycle can handle all those bumpy rides without turning into a twisted pretzel? The secret lies, in part, within the fascinating world of interstitial alloys. And while it might sound like something straight out of a sci-fi movie, understanding the basic principle behind it can be surprisingly rewarding, even if you're not a materials scientist!
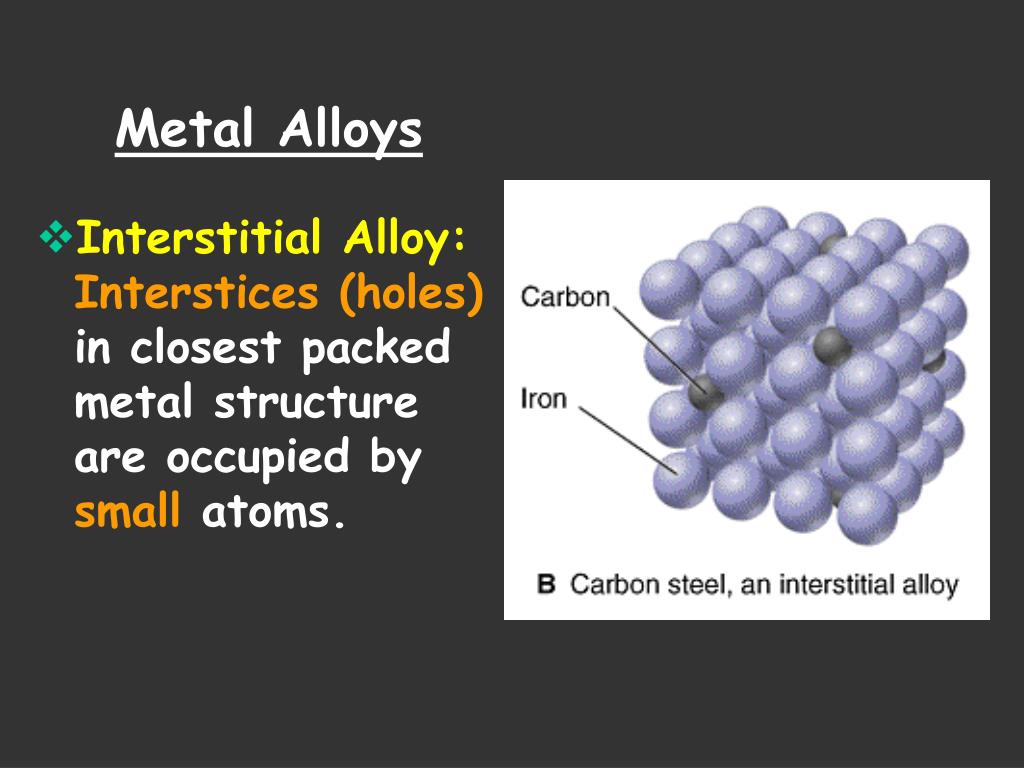
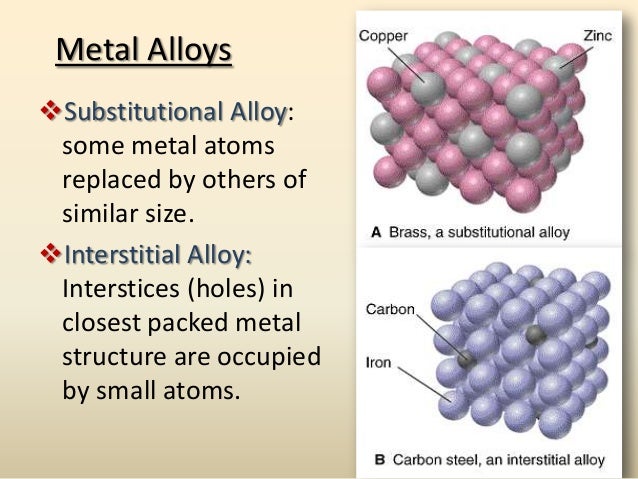
At its core, an interstitial alloy is formed when small atoms, like carbon, nitrogen, or hydrogen, sneak into the spaces (or interstices) between the larger atoms in a metal lattice, like iron. Think of it like jamming tiny pebbles into the gaps between neatly stacked bowling balls. Now, why does this seemingly insignificant addition make such a big difference in the metal's malleability – its ability to be hammered or rolled into thin sheets? Well, that's what we're here to explore.
Artists and hobbyists often unknowingly benefit from the properties of interstitial alloys. Crafters using steel wire for intricate sculptures or jewelry making rely on its enhanced strength and durability. The addition of carbon to iron, creating steel, dramatically increases its resistance to deformation, allowing artists to create more complex and lasting pieces. Imagine trying to build a detailed miniature model out of pure iron – it would be far too soft and easily bent out of shape! Understanding the influence of these small atoms can empower you to choose the right materials for your project, leading to better results and more satisfying creations.
Must Read
For the casual learner, understanding why interstitial alloys are less malleable unlocks a deeper appreciation for the materials that shape our world. From the stainless steel cutlery we use every day to the high-strength alloys used in aerospace engineering, these seemingly simple modifications at the atomic level have profound consequences. The tiny carbon atoms disrupt the smooth, sliding movement of the iron atoms, making it harder for the metal to deform permanently. This increased strength comes at the cost of malleability. Think of it like trying to rearrange a stack of books glued together versus a stack of loose books – the glued stack is stronger, but much harder to manipulate.
Examples abound! Different types of steel, each with varying carbon content, offer a range of properties. High-carbon steel is extremely hard and used for tools like knives, while low-carbon steel is more malleable and used for car bodies. The Eiffel Tower, a testament to 19th-century engineering, is made from wrought iron, a type of iron with a very low carbon content (though not technically an interstitial alloy in the strictest sense, it showcases the impact of controlling the presence of other elements). You can even observe the effects indirectly by comparing the "give" in different types of metal spoons or forks you have at home. Some will bend easily, while others will remain rigid.

While you can't exactly "make" an interstitial alloy at home without a sophisticated lab, you can experiment with heat-treating readily available materials. Annealing (heating and slowly cooling) steel wire can make it softer and easier to bend, while quenching (rapidly cooling) can make it harder and more brittle. Always use proper safety precautions, including heat-resistant gloves and eye protection, and consult reliable sources before attempting any heat-treating experiments. Even just observing the difference in how different metals behave under stress can be a fascinating learning experience.
Ultimately, the beauty of understanding interstitial alloys lies in appreciating the intricate dance of atoms and their profound impact on the macroscopic world. It's a reminder that even the smallest changes can have significant consequences, and that science is all around us, waiting to be explored. It's enjoyable because it's about unraveling a mystery, connecting seemingly disparate concepts, and gaining a deeper appreciation for the materials that shape our everyday lives. So, next time you see a sturdy steel structure, take a moment to appreciate the tiny atoms that are working tirelessly to keep it strong!