Which Property Do Transition Metals Have In Common

Ever wonder what connects that shiny piece of jewelry you adore with the catalytic converter in your car? Or perhaps how the vibrant colors in fireworks relate to the strength of steel bridges? The answer lies in a fascinating group of elements: the transition metals.
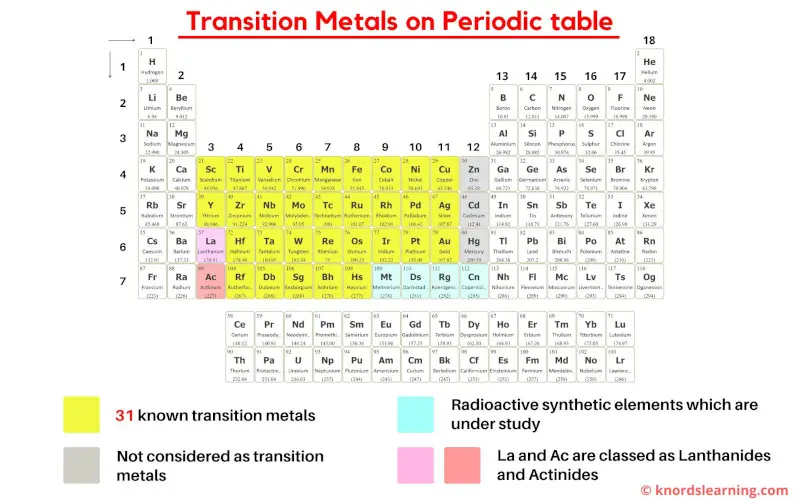
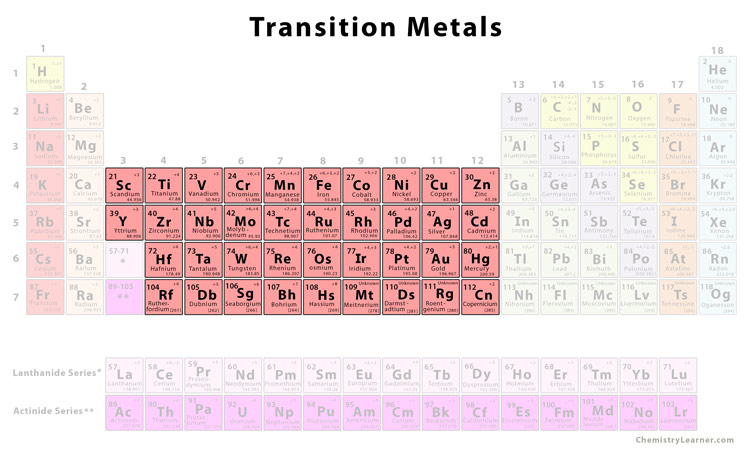
They're All About "D"
Now, before your eyes glaze over, let's ditch the textbook jargon. Imagine the periodic table like an apartment building. Transition metals are the cool kids living in the "d-block" apartments. What they share? An unfinished business with their "d" orbitals - a specific kind of electron holding space.
This unfinished business is the secret sauce to their shared properties. It’s like having a partially completed Lego set; the possibilities are endless!
Must Read
Colorful Characters
One of the most noticeable things about transition metals is their penchant for vibrant colors. Think of the deep blue of copper sulfate crystals, the green of nickel compounds, or the purple of amethyst (which gets its color from iron impurities). It's like nature’s own rainbow factory.
This color show happens because those "d" electrons can jump between energy levels when exposed to light. The specific wavelengths of light they absorb determine the color we see. It’s like a tiny electron dance party!
This is why you see them in pigments, dyes, and even stained glass. It makes them the artists of the periodic table.

Masters of Disguise (Variable Oxidation States)
Transition metals are chameleons. They can exist in multiple oxidation states, which is a fancy way of saying they can form different numbers of bonds with other atoms. They're adaptable, like social butterflies at a party.
Iron, for example, can be Fe2+ or Fe3+. This ability to change their "charge" is crucial for many biological processes, like the way your blood carries oxygen. Pretty important for staying alive!
It also makes them superb at catalyzing reactions, which brings us to….
The Catalytic Converters
Many transition metals are excellent catalysts. A catalyst speeds up a chemical reaction without being consumed in the process. Think of them as matchmakers for molecules.
Platinum, palladium, and rhodium are used in catalytic converters in cars to reduce harmful emissions. They help convert nasty gases like carbon monoxide into less harmful substances. A small amount can clean up a lot of mess!
Iron is also a key catalyst in the Haber-Bosch process, which is used to produce ammonia for fertilizers. This single process has had a massive impact on global food production. Talk about powerful!
Magnetic Personalities
Some transition metals, like iron, nickel, and cobalt, are famously magnetic. This is due to the unpaired electrons in their "d" orbitals aligning and creating a strong magnetic field.
This is why magnets stick to your refrigerator. They allow us to store data on hard drives. Plus, who doesn't love playing with magnets?
/metals-including-clockwise-from-left-copper-aluminium-zinc-iron-and-lead-74102855-57ced3d33df78c71b643079f.jpg)
The strength of the magnetism can also depend on temperature. Heat it up enough, and the magnetism disappears!
Strong and Stable (Alloys)
Transition metals often form strong and stable alloys when mixed with other metals. Steel, for example, is an alloy of iron and carbon (and sometimes other elements like chromium or nickel).
Alloys are often stronger and more resistant to corrosion than the individual metals. That's why they are used in everything from bridges and buildings to airplanes and cars. Strength in numbers, right?
Adding even a small amount of a transition metal can dramatically change the properties of the original metal.
They’re Everywhere!
From the iron in your blood to the titanium in artificial joints, transition metals are essential for life and technology. They’re hiding in plain sight.
Next time you see a colorful piece of jewelry, a sturdy steel structure, or a sparkling firework, remember the humble transition metals. They are the unsung heroes of the periodic table.
They’re not just elements, they're elements of surprise, beauty, and functionality. They are the foundation of so many things we take for granted.
A Few Fun Facts
- Gold (Au) and Silver (Ag) are highly valued for their resistance to corrosion. That’s why they’re used in jewelry and electronics.
- Titanium (Ti) is incredibly strong and lightweight, making it perfect for airplanes and prosthetics.
- Zinc (Zn) is essential for a healthy immune system. It's often found in supplements.
- Vanadium (V) gets its name from Vanadis, the Scandinavian goddess of beauty.
- Manganese (Mn) is important for bone health and wound healing.
So next time you gaze at the periodic table, give a nod to the transition metals. They’re the busy bees, the colorful artists, and the sturdy builders of our world. They are definitely doing their part.
