Which Metal Has Highest Thermal Conductivity

Ever wondered why your phone gets hot when you're playing a graphics-intensive game? Or why some pots and pans heat up incredibly quickly, while others take forever? The answer lies in thermal conductivity, a fascinating property of materials that dictates how easily heat flows through them. And when it comes to metals, the race for the highest thermal conductivity is a surprisingly exciting one, with real-world implications for everything from electronics cooling to cookware design. It's a topic that touches our lives every day, even if we don't realize it!
So, what exactly is thermal conductivity, and why should we care? Simply put, it's a measure of how well a material conducts heat. A material with high thermal conductivity will transfer heat rapidly, while one with low thermal conductivity will act as an insulator, slowing down heat transfer. Understanding this property is crucial for numerous applications. Think about designing efficient heat sinks for computers to prevent overheating, or choosing the right material for a cooking pot that distributes heat evenly and quickly. Even building insulation relies on materials with low thermal conductivity to keep our homes warm in winter and cool in summer.
Now, for the big reveal! The metal with the highest thermal conductivity at room temperature is silver. Yes, that shiny, precious metal often associated with jewelry and silverware reigns supreme in the world of heat transfer! It boasts a thermal conductivity of around 429 Watts per meter-Kelvin (W/mK). That means it can move a lot of heat, very quickly.
Must Read
But why aren't we using solid silver cookware and computer components everywhere? The answer, unsurprisingly, is cost. Silver is an expensive metal, and using it on a large scale would be economically prohibitive for most applications. That's where other metals come into play.
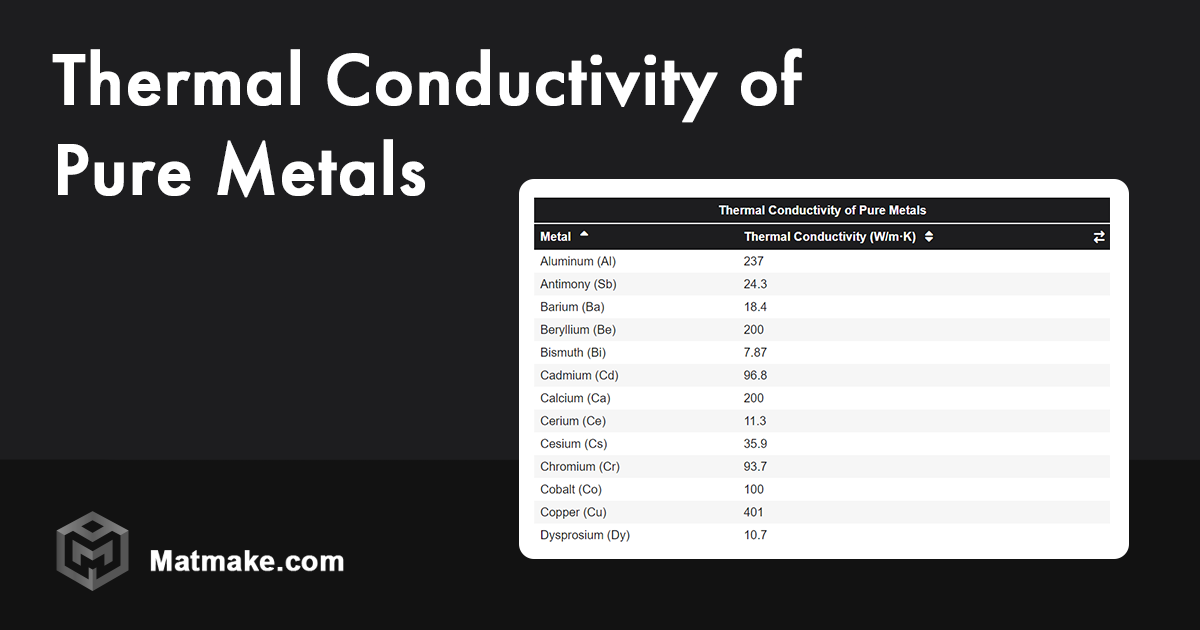
While silver takes the gold (or should we say, the silver?) other metals offer a good balance of thermal conductivity and cost-effectiveness. Copper, with a thermal conductivity of around 401 W/mK, is a close second to silver and is far more affordable. This is why copper is widely used in electrical wiring, heat sinks, and cookware. Gold, another excellent conductor at around 317 W/mK, finds use in specialized electronic applications where high reliability and resistance to corrosion are critical. Aluminum, at around 237 W/mK, is also popular due to its lightweight nature and relatively good thermal conductivity, making it ideal for applications like car radiators and some heat sinks.
In conclusion, while silver holds the title for the highest thermal conductivity among metals, practical considerations like cost and availability often lead to the use of other materials like copper and aluminum. Understanding the principles of thermal conductivity helps us design better products, improve energy efficiency, and even keep our gadgets from overheating. So, the next time you feel the warmth of your phone or admire a well-designed cooking pot, remember the amazing property of thermal conductivity and the fascinating world of metals that make it all possible!