Which Ion Has The Largest Radius

Ever wondered about the ultimate showdown of the atomic world? It's not about explosions or fancy gadgets. It's about size! We're diving into the quirky question: Which ion is the biggest?
Ions: Atoms with Attitude
First, let's talk ions. Imagine atoms as tiny Lego bricks. Normally, they're neutral, with the same number of positive and negative bits.
But sometimes, atoms are greedy! They either steal or give away electrons. This makes them charged particles called ions. Think of them as atoms with attitude!
Must Read
If an atom gains electrons, it becomes a negative ion (anion). If it loses electrons, it becomes a positive ion (cation). This gain or loss changes their size!
Why Size Matters
Size isn't everything, right? Well, in the ion world, it's pretty darn important! The size of an ion influences how it interacts with other ions. This plays a vital role in forming compounds.
It affects how chemicals react and what structures they can build. Ionic radius is vital for understanding chemical behavior.
Imagine trying to fit puzzle pieces together. A big ion is a big puzzle piece! It needs a corresponding space to fit comfortably.
The Size Contest: Who's the Winner?
So, who takes home the "Biggest Ion" trophy? It's not a simple answer! Several factors influence ionic size.
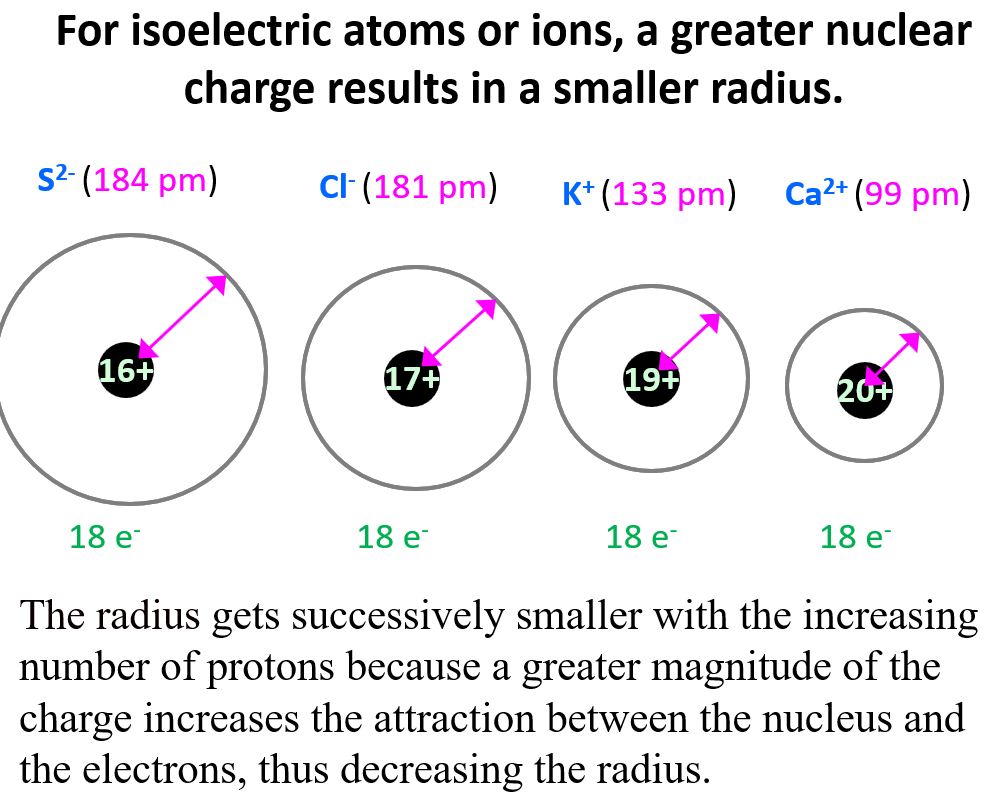
The nuclear charge (positive power in the atom's center) pulls the electrons in. More protons, more pull, smaller size. Simple enough?
The number of electrons also plays a role. More electrons mean more repulsion. This "electron cloud" expands, making the ion bigger. Things are getting interesting!
Negative Ions are Usually the Champions
Generally, negative ions (anions) are larger than positive ions (cations). Anions gain electrons. That extra electron cloud causes the ion to expand.
Cations, on the other hand, lose electrons. This loss reduces electron repulsion and exposes the remaining electrons to a stronger pull from the nucleus. The ion shrinks!
Think of it like this: adding air to a balloon (anion) vs. deflating a balloon (cation). Which one gets bigger?

The Periodic Table's Role
The periodic table can give you some clues! As you move down a group (column), the ions generally get larger. This is because you're adding more electron shells. More shells, bigger ion!
But it's not always that straightforward. The trend can be affected by other factors such as electron-electron repulsion.
This is why knowing the trends on the periodic table is useful. It guides us towards the larger atoms.
Examples of Big Ions
So, which specific ions are likely contenders? Large anions like iodide (I-) and telluride (Te2-) are strong candidates. They have many electrons and relatively weak nuclear attraction compared to the number of electrons.
Think of those giant, fluffy clouds in the sky – these ions are kind of like that!

But it's a relative thing. Comparing ions in the same period or group is the best way to get a true comparison.
Why is This Entertaining?
Okay, maybe "entertaining" is a strong word! But there's something oddly satisfying about understanding these tiny building blocks of the universe.
It's like solving a miniature puzzle. Understanding the rules that govern them is really cool!
Plus, it reminds us that everything around us is made of these tiny things. They all follow the rules of chemistry and physics. It's mind-blowing when you think about it.
Dive Deeper!
Want to learn more? Explore the wonderful world of chemistry textbooks! Look at online resources about ionic radii trends.

You can find charts comparing the sizes of different ions. Become an ion size expert!
Who knows, you might just discover the next record-breaking ion! Keep searching and keep learning.
Size Doesn't Always Matter... But It's Interesting!
The quest for the largest ion is a reminder that even the tiniest things can be fascinating. It opens our eyes to the intricate rules that govern the atomic world. So go ahead, delve into it yourself!
While size isn't everything in chemistry, it sure is an interesting property to explore. Understanding these trends opens doors for further learning!
Happy exploring! The atomic world awaits your curiosity.
