Which Equation Represents A Neutralization Reaction
Okay, buckle up buttercups! We're about to dive into the splashy world of neutralization reactions. Think of it as a superhero showdown, but instead of capes and tights, we've got acids and bases!
It’s like the ultimate power couple balancing each other out. No drama, just sweet, sweet neutrality!
The Acid-Base Tango: Finding the Perfect Match
So, how do we spot a neutralization reaction equation amidst the chaotic soup of chemical formulas? Fear not, my friends, for I'm about to spill the tea (or, you know, the chemical solution!).
Must Read
It's all about finding the equation where an acid and a base meet and decide to chill out together, resulting in the formation of salt and water.
Let's break it down, shall we? Imagine it like this: Acid is a tiny little devil, base is an angel. They meet. Instead of war, they realize they actually complement each other and then boom, they create something brand new!
Acid: The Sourpuss
First up, we have the acids. Think of them as the sour patch kids of the chemical world. They donate protons (H+), because, let's be honest, who doesn't love protons?
They're the life of the party! Common acids include hydrochloric acid (HCl) – the stuff in your stomach that helps digest food – and sulfuric acid (H2SO4) – a super-powerful industrial acid.
Acids are those substances that can taste sour. Avoid tasting them in lab though!
Base: The Bitter Buddy
Now, let's bring in the bases. These are the unsung heroes who can accept those protons with open arms! They are often bitter.
Think of bases as the calming influence, always there to balance things out. A classic example is sodium hydroxide (NaOH), also known as lye – used in soap making and cleaning.
Another common base is ammonia (NH3), which you might recognize from household cleaners. Bases are so good at accepting protons, it's almost suspicious!
Spotting the Neutralization Equation: The Big Reveal
Alright, let's get down to business. How do you identify a neutralization reaction equation? Look for this magical formula:
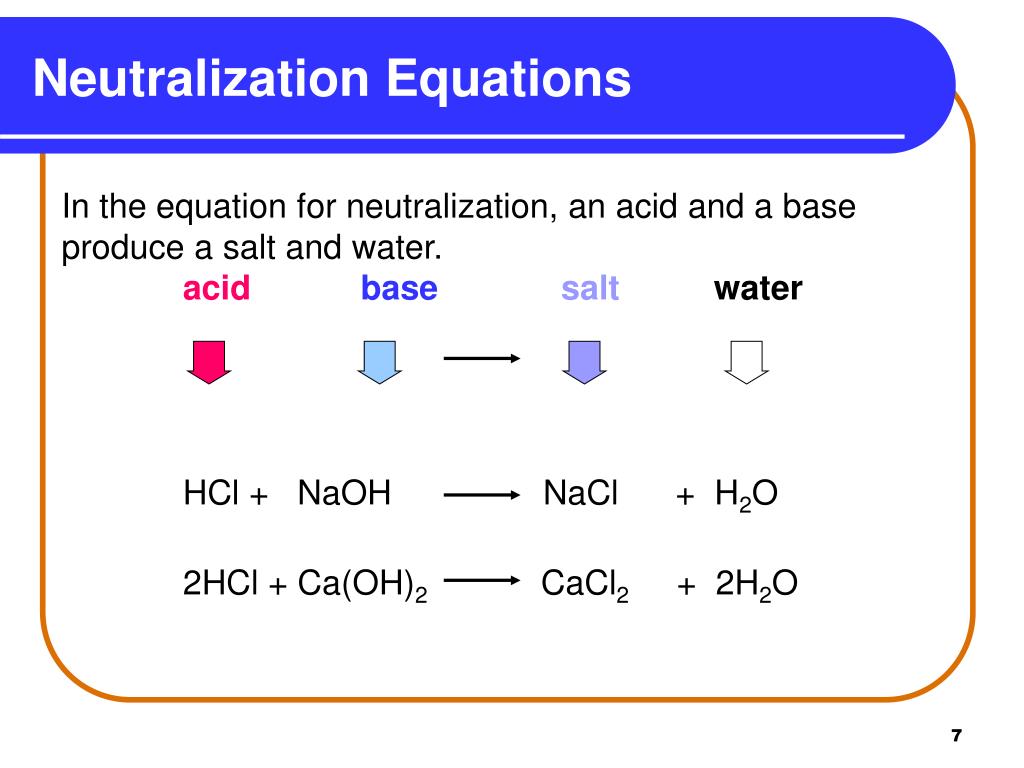
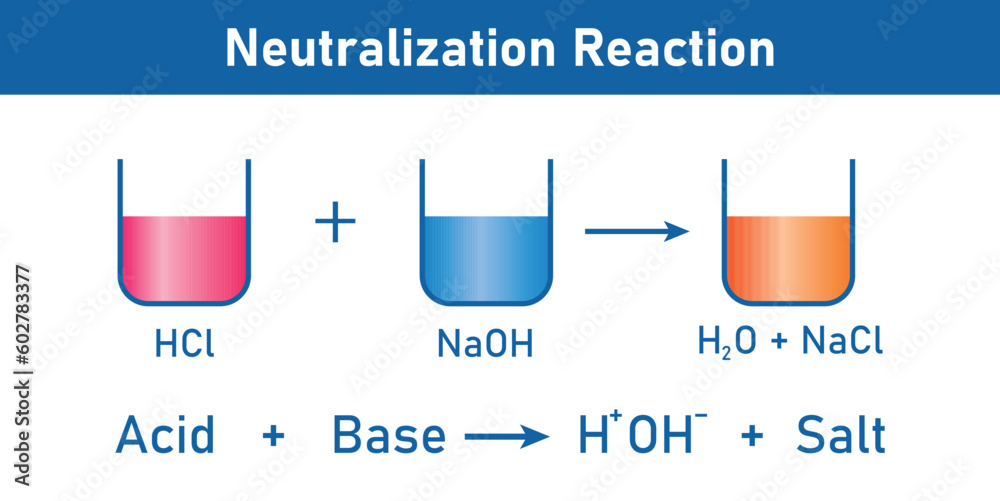
Acid + Base → Salt + Water
It's that simple! If you see an acid and a base reacting to form a salt and water, congratulations, you've found a neutralization reaction.
Let's look at some examples, because nothing makes things clearer than examples! I am so excited to share with you all!
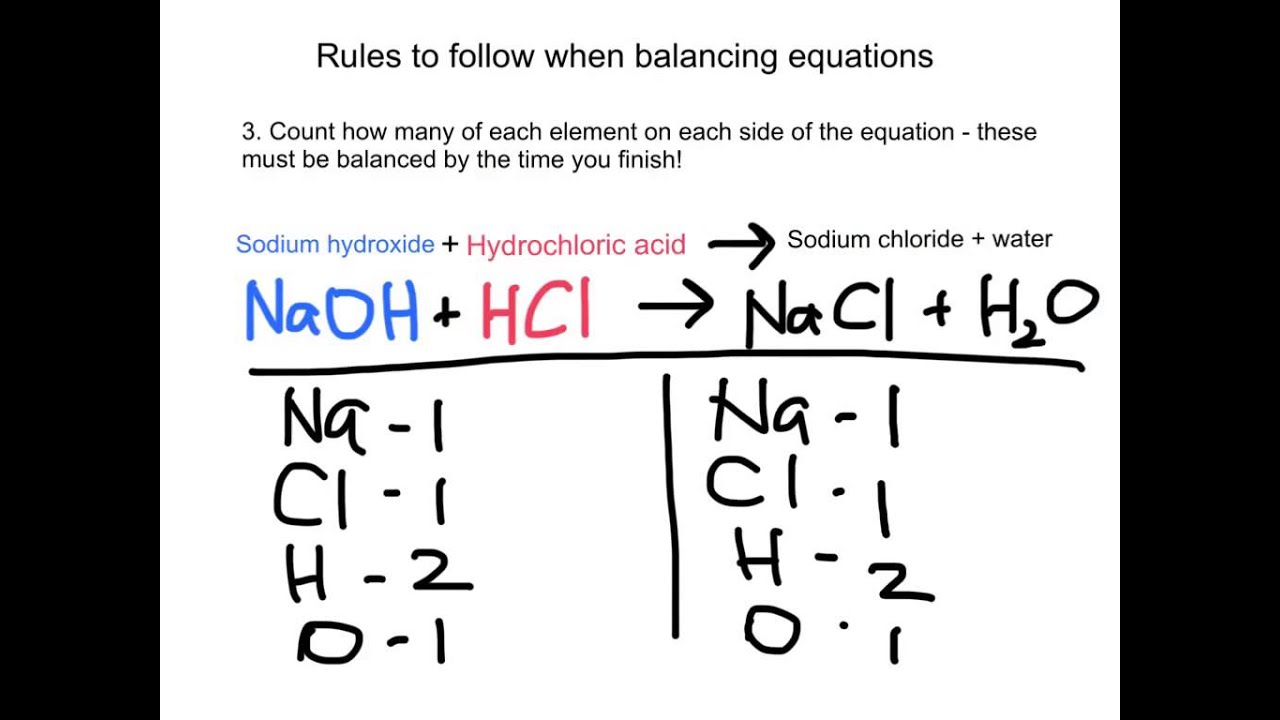
Example 1: Hydrochloric Acid and Sodium Hydroxide
Consider this reaction: HCl (acid) + NaOH (base) → NaCl (salt) + H2O (water). Ta-da! Hydrochloric acid (HCl) reacts with sodium hydroxide (NaOH) to produce sodium chloride (NaCl), which is just fancy name for table salt and water.

This is the quintessential neutralization reaction. It’s like the Romeo and Juliet of chemistry, except without all the tragic dying at the end. They are just balancing out!
Everyone lives happily ever after, in a neutral, slightly salty paradise.
Example 2: Sulfuric Acid and Potassium Hydroxide
Another one! Take this reaction: H2SO4 (acid) + 2KOH (base) → K2SO4 (salt) + 2H2O (water). Here, sulfuric acid (H2SO4) reacts with potassium hydroxide (KOH) to form potassium sulfate (K2SO4) and water.
Notice how we need two molecules of KOH to neutralize one molecule of H2SO4? That's because sulfuric acid is a "diprotic" acid, meaning it donates two protons instead of one.
It's like it's twice as generous! Balancing these equations is crucial to knowing just how much of each ingredient you need for the perfect chemical cocktail.
Example 3: Acetic Acid and Sodium Bicarbonate
Even something familiar can be a neutralization reaction! Have you ever mixed vinegar (acetic acid) and baking soda (sodium bicarbonate)? CH3COOH (acid) + NaHCO3 (base) → CH3COONa (salt) + H2O (water) + CO2 (gas)
You've probably seen it erupt like a mini-volcano! That fizzing action is due to the carbon dioxide gas (CO2) that's released as a byproduct. While there is a gas involved, but it is still a neutralization reaction.
The reaction creates sodium acetate, water and carbon dioxide. It's a classic science fair experiment, and now you know the chemistry behind it.
When It's NOT a Neutralization Reaction
Now, let's talk about what doesn't qualify. Not every reaction involving an acid or a base is a neutralization reaction.
For example, consider this reaction: HCl + Zn → ZnCl2 + H2. Hydrochloric acid reacts with zinc to produce zinc chloride and hydrogen gas.
While it involves an acid, there's no base involved, and no water is formed. It's a single replacement reaction, not a neutralization reaction.
Similarly, combustion reactions, precipitation reactions, and redox reactions aren't neutralization reactions either, even if they involve acidic or basic conditions.
It's all about that specific "acid + base → salt + water" formula.

Why Should You Care? The Awesome Applications
So, why should you care about neutralization reactions? Well, aside from being super cool (which they are!), they have tons of practical applications.
Think about antacids you take when you have heartburn. Those are bases that neutralize the excess stomach acid causing you discomfort!
Farmers use neutralization reactions to adjust the pH of soil, making it more suitable for growing crops. Waste water treatment plants use this to make waste water safe for the environment.
From treating bee stings (which are acidic, so you use a base like baking soda) to cleaning up chemical spills, neutralization reactions are essential in many areas.
A Final Word of Encouragement
So, there you have it! The not-so-secret code to identifying neutralization reactions. Remember the formula: Acid + Base → Salt + Water.
Keep your eyes peeled for acids and bases teaming up to create salt and water, and you'll be a neutralization reaction ninja in no time!
Happy reacting, my friends!
