Which Element Would Have The Lowest Electronegativity

Hey everyone! Ever wondered what makes elements tick? I mean, beyond the whole "everything is made of them" thing? Today, we're diving into a property called electronegativity, and figuring out which element is the chillest, most un-clingy of the bunch. And trust me, even if you haven't thought about chemistry since high school, this is surprisingly relevant to your life.
Think of electronegativity like this: It's an element's desire to hog electrons in a relationship (a chemical bond, that is). Some elements are real electron-grabbers, super possessive types. Others? Not so much. They're more like, "Hey, you do you with those electrons, friend!"
So, Why Should I Care?
Okay, fair question. Why should you, a perfectly reasonable human being just trying to get through the day, care about which element has the lowest electronegativity? Well, it affects everything from the way molecules form to the properties of the materials around you. Imagine trying to bake a cake if some ingredients were violently attracted to each other while others just politely ignored each other. You'd end up with a lumpy mess!
Must Read
Plus, understanding electronegativity helps us understand why some things are conductors of electricity (like copper wires – those guys are practically giving electrons away!) and why others are insulators (like the rubber coating – holding onto electrons for dear life!). It's like understanding the personality types in your friend group – knowing who's outgoing and who's shy helps you predict how they'll act at a party, right?
The Usual Suspects: Metals on the Left
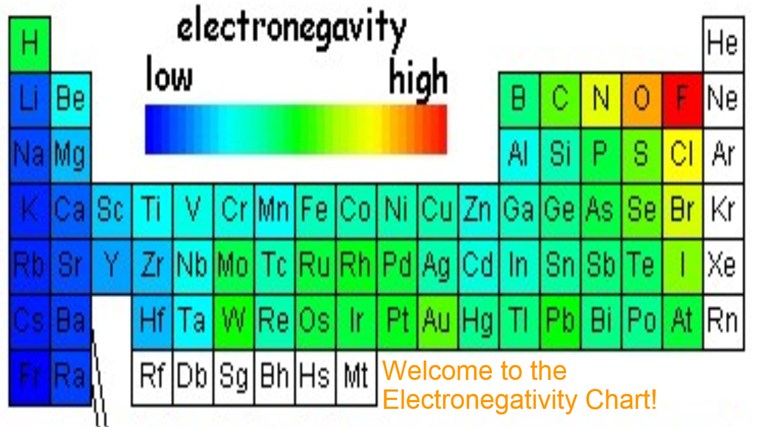
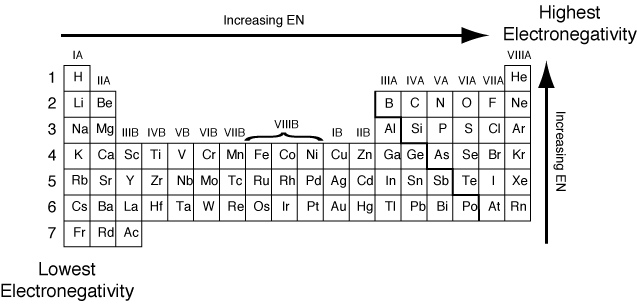
Now, where do we even start looking for the least electronegative element? Well, on the periodic table, electronegativity generally increases as you go from left to right and from bottom to top. This means the elements on the left and towards the bottom are more likely to be the relaxed, "go with the flow" types.
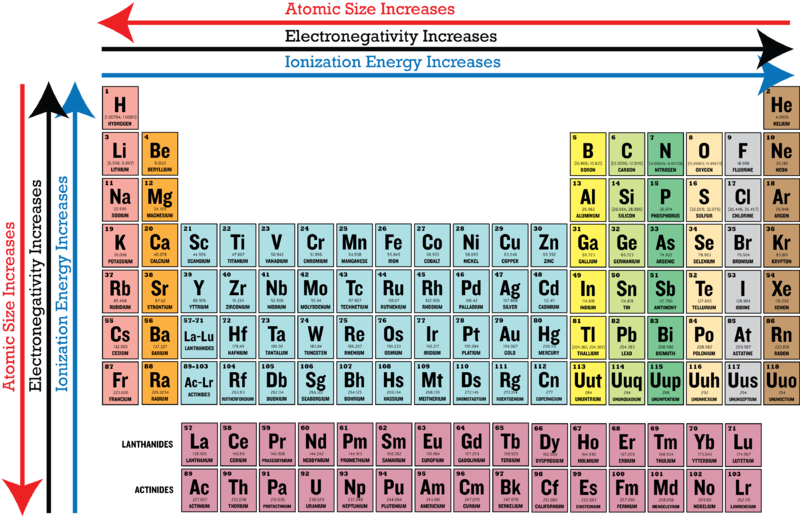
We're generally talking about metals here. Remember, metals like to lose electrons to form positive ions. That's the opposite of being electronegative! They're more like, "Take these electrons, please! I have too many."
And the Winner Is... Francium! (Probably)
Drumroll, please! The element generally considered to have the lowest electronegativity is Francium (Fr). It's an alkali metal, sitting way down in the bottom left corner of the periodic table. Francium is like the ultimate electron-donor, the Mother Teresa of the element world (if Mother Teresa gave away electrons instead of, you know...).
Now, I said "probably" because Francium is super rare and radioactive. It's not exactly something we can easily experiment with. It’s more like a legend, whispered about in chemistry labs.
Think of it this way: Francium is like that super rare, vintage comic book that everyone assumes is worth a fortune, but no one has actually seen one for sale in decades. We theoretically know it’s valuable (or, in this case, not electronegative), but it's hard to prove definitively.
Cesium: A Close Second and More Accessible
Because Francium is so elusive, sometimes Cesium (Cs), which is right above it on the periodic table, gets the nod as the most practically electropositive (least electronegative) element. Cesium is still pretty reactive, but it's a lot more stable and easier to study than Francium. So, in the real world, Cesium often takes the crown.
Consider Cesium your reliable, friendly neighbor who's always willing to lend you a cup of sugar (or, in this case, an electron). It's not quite as "legendary" as Francium, but it's a solid, dependable choice.
Why Does This Even Happen?
Okay, a quick bit of science: Atoms with fewer electrons in their outer shell (valence electrons) and with those electrons further away from the nucleus (the positively charged center of the atom) have a lower attraction to those electrons. Francium and Cesium both have only one valence electron, and those electrons are shielded from the nucleus by many other electron shells. This makes it easy for them to lose that electron and achieve a stable electron configuration.

Think of it like trying to hold onto a slippery watermelon with one hand while wearing oven mitts. It's just not gonna happen! The watermelon (valence electron) is too far away (shielded by other electron shells) and your grip is weak (low electronegativity).
Electronegativity in Your Everyday Life
So, there you have it! Francium (or Cesium) takes the prize for lowest electronegativity. And while you might not be thinking about Francium every day, understanding this concept helps you understand the world around you. It explains why some materials bond the way they do, why some conduct electricity, and even why water is such a unique and life-sustaining substance.
Next time you're cooking, building something, or even just flipping a light switch, take a moment to appreciate the amazing dance of electrons that makes it all possible. And remember the chill, un-clingy Francium, chilling at the bottom of the periodic table, not wanting to steal any electrons from anyone.
