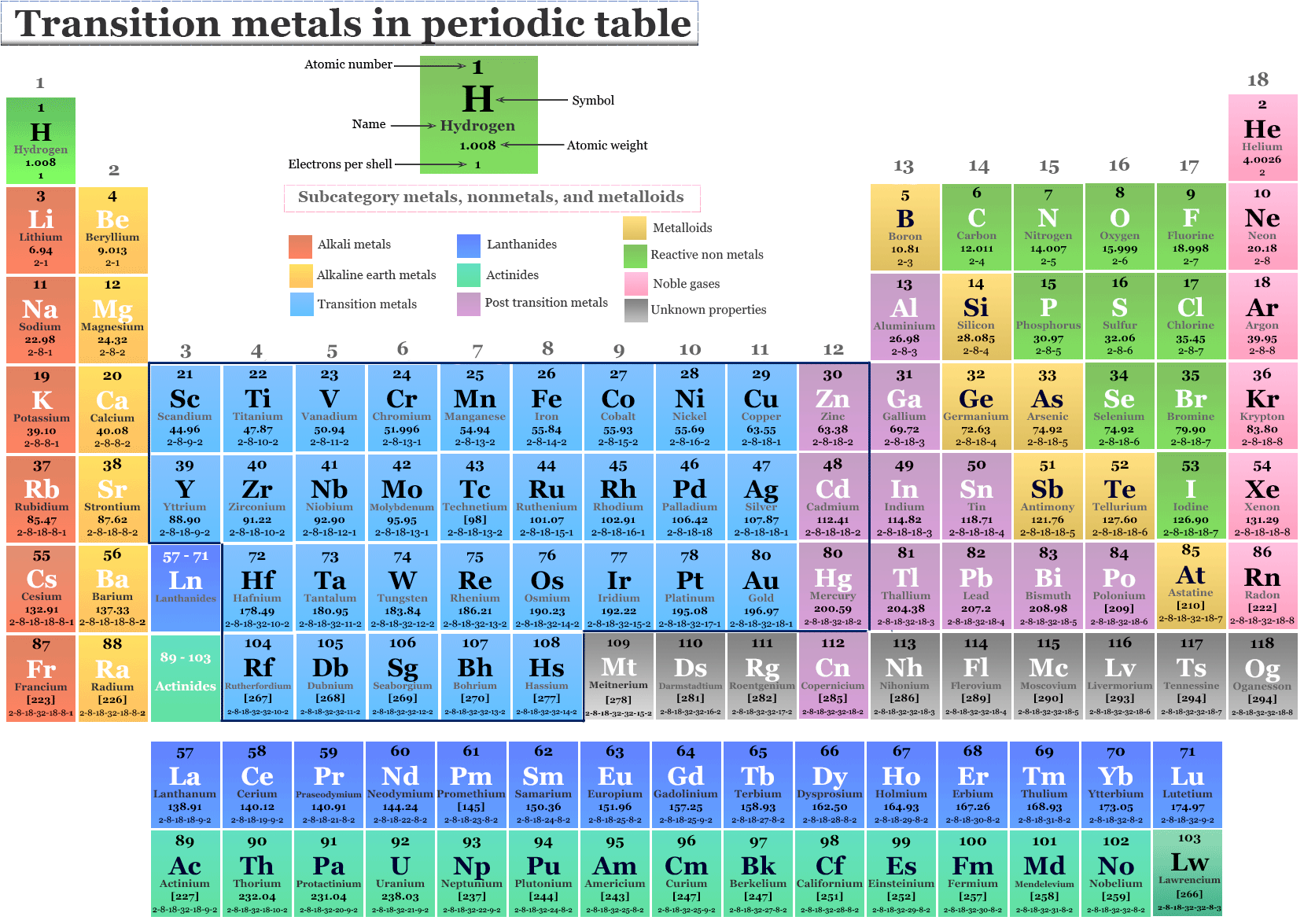
Where Are Transition Metals On The Periodic Table
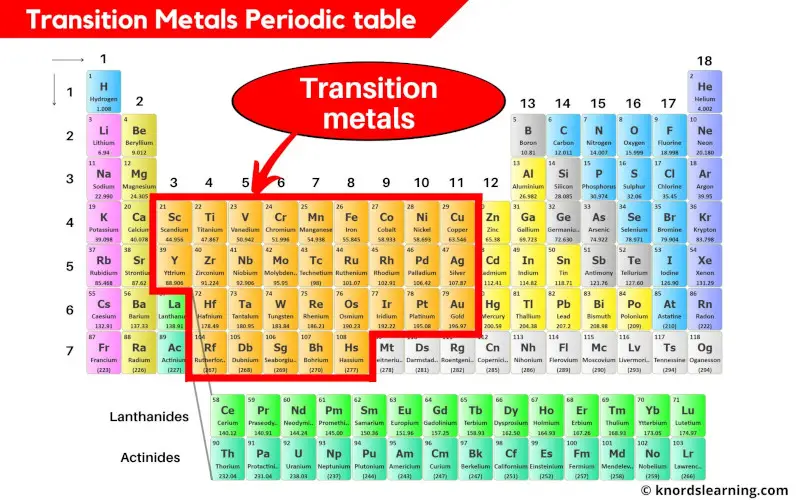
Ever glanced at the periodic table and thought, "Wow, that's...a lot?" You're not alone! It's like a giant, organized Lego set of all the elements. And right smack-dab in the middle, like the cool centerpiece of a chemistry party, are the transition metals.
But where exactly are these metallic marvels hanging out? Think of the periodic table as a neighborhood. On the left, you've got the super-friendly alkali and alkaline earth metals. On the right, the sometimes standoffish nonmetals and the noble gases, who are basically the celebrities of the element world, too cool to react with anyone (usually!).
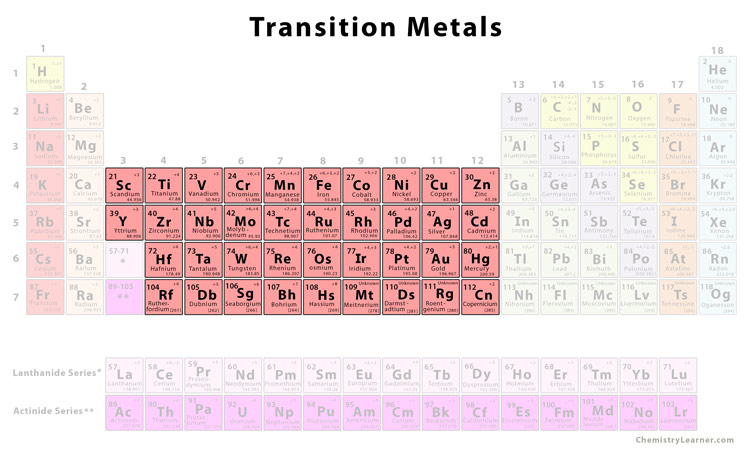
The transition metals? They're in the D-block, chilling in the central block of the periodic table, wedged between Groups 2 and 13. Imagine them as the adaptable kids on the block, able to play well with pretty much everyone.
Must Read
Why Are They So Special?
These aren't your average metals. Sure, they're shiny, good conductors of electricity, and generally tough cookies. But it's their personality that makes them truly fascinating. They're the chameleons of the element world, able to change their oxidation states (basically their "charge") and form all sorts of colorful compounds.
Think of iron (Fe). It's the backbone of our infrastructure, making up bridges, buildings, and even the red blood cells that keep us alive. Then there's copper (Cu), wiring our homes and turning statues green with a touch of time and weather. And let's not forget gold (Au), the symbol of wealth and beauty throughout history.
But it's not just their practical applications. Transition metals are responsible for some truly stunning colors. The vibrant blue of some gemstones? Often due to cobalt (Co). The deep purple of amethyst? Thank manganese (Mn). They're like the artists of the atomic world, painting our world with vibrant hues.
More Than Meets the Eye
What makes them so flexible? It all comes down to their electron configurations. Without diving too deep into the science-y stuff, they've got electrons in their outer shells that are a bit more… available. These electrons can jump around and form different bonds, leading to their diverse oxidation states and colorful compounds.
They are also excellent catalysts. Catalysts are substances that speed up chemical reactions without being consumed themselves. Many industrial processes rely on transition metals as catalysts to produce everything from plastics to pharmaceuticals. Think of them as tiny matchmakers, bringing other elements together to create something new and useful.

Want to see them in action? Just look around! Your stainless-steel cookware? Contains chromium (Cr) for corrosion resistance. The rechargeable batteries in your phone? Likely use nickel (Ni) and cadmium (Cd). Even the pigments in your favorite paints probably contain transition metal compounds.
"The periodic table is a map of the chemical world, and the transition metals are a crucial part of that map."
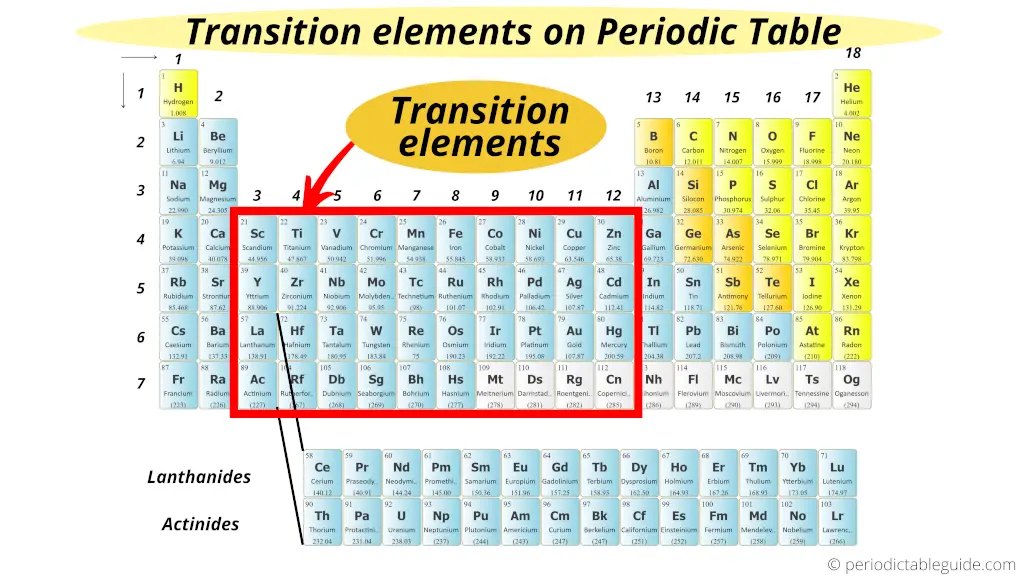
Time to Explore!
So, the next time you glance at the periodic table, take a moment to appreciate the transition metals. They're not just a bunch of elements sitting in the middle; they're the workhorses, the artists, and the catalysts of the chemical world. They are involved in so many processes and items that we use every day, they are truly indispensable.
Go ahead, grab a periodic table (they're easily found online) and take a closer look at the D-block. You might be surprised at just how many familiar faces you find. Maybe you’ll even discover a new favorite element! Who knows, exploring these metallic marvels might just spark your inner chemist!
They are pretty cool elements. Have fun exploring them!