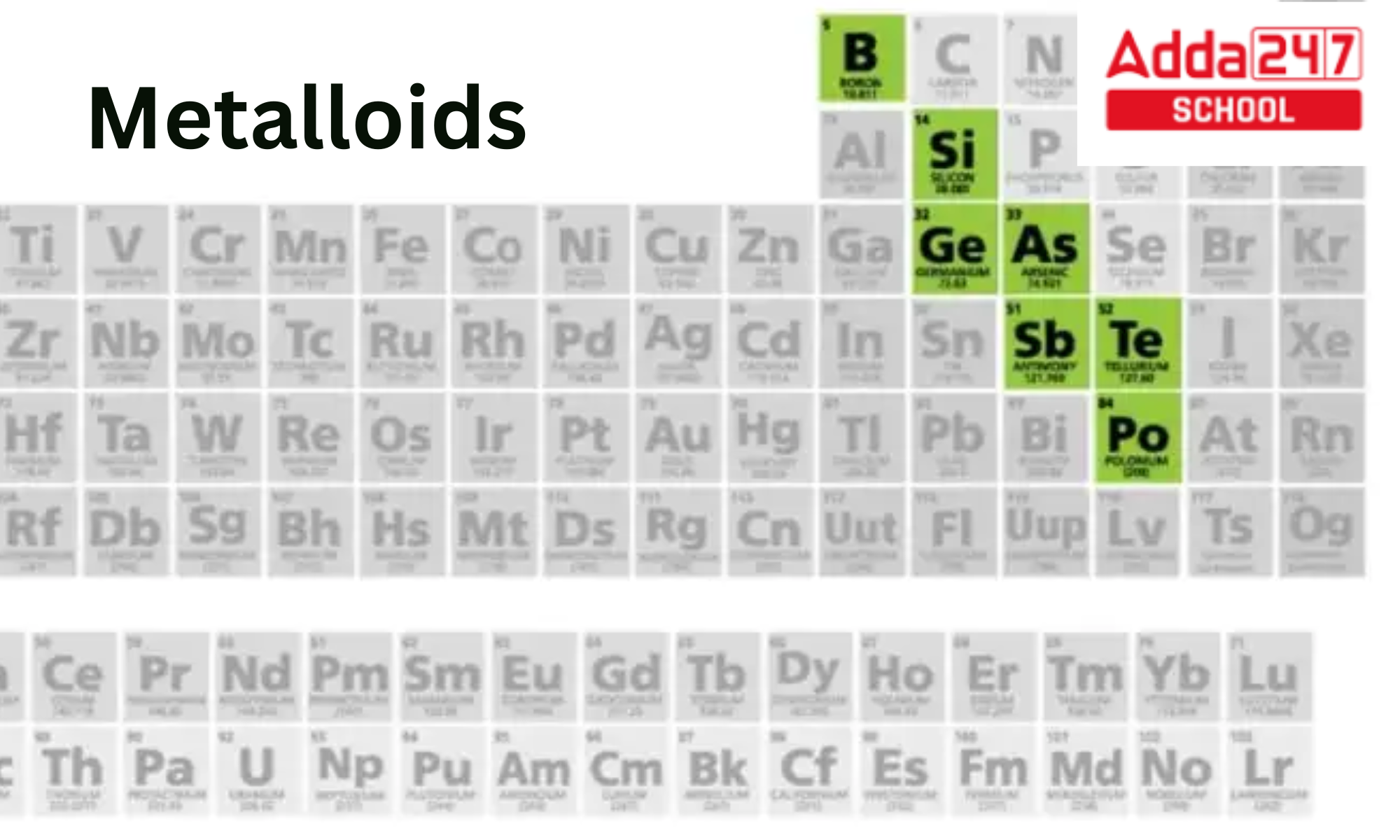
Where Are The Metalloids Found On The Periodic Table

Hey everyone! Ever looked at the periodic table and thought, "Wow, that's a lot of squares?" Yeah, me too! But have you ever zoomed in on those elements chilling out in between the metals and nonmetals? I'm talking about the metalloids! They're like the chameleons of the element world, and today we're going to explore where these interesting guys hang out.
Think of the periodic table as a neighborhood. On one side, you've got the super outgoing, always-ready-to-react metals. On the other side, the nonmetals, a bit more reserved, maybe a little picky about who they interact with. And right there in the middle, acting as the bridge between these two very different groups? Our metalloids! So, where exactly are they? Let's find out!
The Metalloid Border Patrol
You'll find the metalloids clustered along a diagonal line, kind of like a staircase, on the right-hand side of the periodic table. It's not a perfectly straight line, mind you, more like a slightly tipsy one. We’re talking about elements like Boron (B), Silicon (Si), Germanium (Ge), Arsenic (As), Antimony (Sb), Tellurium (Te), and sometimes Polonium (Po) and Astatine (At) get a little metalloid love. Think of them as the gatekeepers of reactivity.
Must Read
But why are they so interesting? Well, they're not quite metals, and they're not quite nonmetals. They have properties of both! It's like they went to the element buffet and took a little bit of everything. What's cooler than that?
Metalloid Superpowers: Not Quite Clark Kent, But Close
What makes these elements so special is their in-between-ness. For example, some metalloids, like Silicon and Germanium, are semiconductors. Sounds fancy, right? It basically means they can conduct electricity, but not as well as metals. They're like the dimmer switch of the element world. You can control how much electricity flows through them. And that's why they're crucial for things like computer chips and solar panels. Imagine trying to run your phone without a semiconductor! Yikes.

Others, like Arsenic and Antimony, can be used in alloys (mixtures of metals) to make them harder or more resistant to corrosion. Think of it as adding a special ingredient to make the final product even better! They're the supporting actors that make the lead shine brighter.
Why This Matters (And Why You Should Care)
So, why should you care where these metalloids are on the periodic table? Because they are everywhere! They're not just some obscure elements locked away in a lab. They're in your phone, your computer, your car, maybe even the medicine you take. They're the unsung heroes of modern technology and industry.
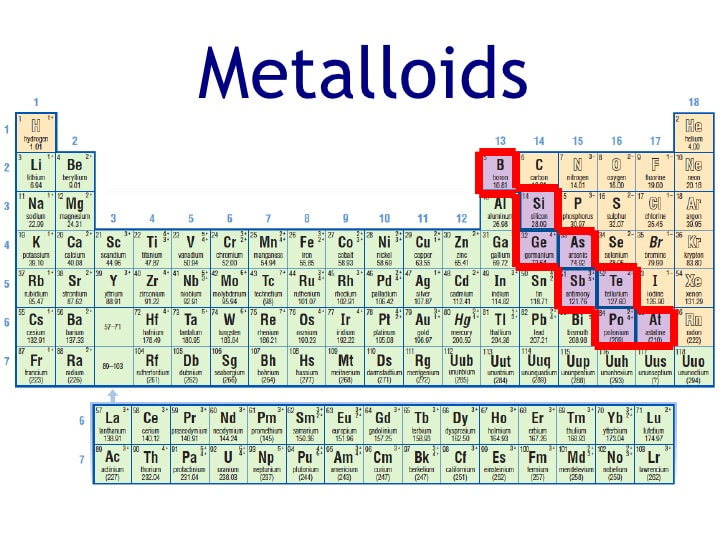
And understanding where they are on the periodic table helps us understand their properties and how they interact with other elements. It’s like knowing the recipe to a delicious dish – knowing the ingredients (elements) and how they combine (react) helps you create something amazing!
Think of it This Way...
Imagine the periodic table as a big party. The metals are the loud, energetic dancers, always up for anything. The nonmetals are the more reserved guests, carefully choosing who they interact with. And the metalloids? They're the social butterflies, mingling with everyone, making connections, and keeping the party flowing smoothly. They're the glue that holds everything together!

So, the next time you glance at the periodic table, take a moment to appreciate the metalloids. They may not be the flashiest elements, but they play a vital role in our world. They're the masters of adaptability, the bridge-builders, and the unsung heroes of the element world. Now, isn't that something to think about?
And hey, maybe you'll even impress your friends with your newfound metalloid knowledge. "Oh, you didn't know that Silicon is a metalloid and is essential for computer chips? Let me tell you..." 😉