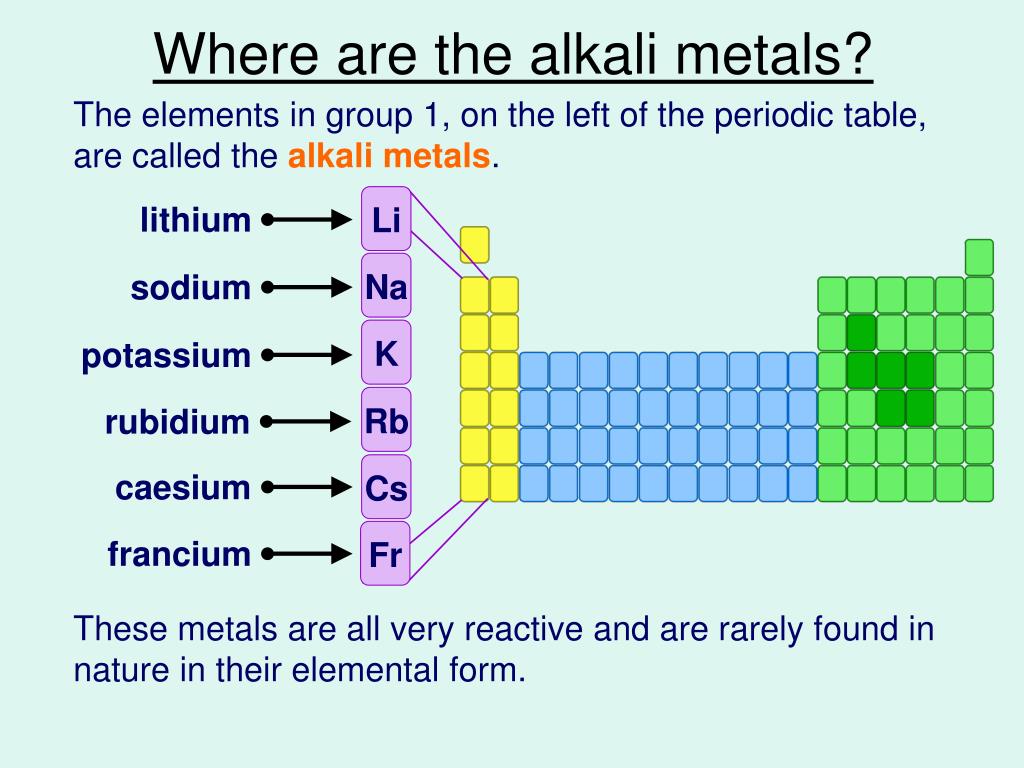
Where Are The Alkali Metals Located
Ever wondered where to find the most outgoing personalities in the periodic table? Well, look no further than the alkali metals! These elements, known for their energetic behavior, are quite the characters.
Hunting for the Hotshots: Where do they hang out?
First things first: You won't find them chilling in their pure form in nature. Imagine trying to keep a group of hyperactive kids in a perfectly still pose – it’s just not happening! They're way too reactive.
Instead, they're usually found hanging out in compounds. They're always eager to bond with other elements, like chlorine and oxygen. Think of them as the ultimate social butterflies of the element world.
Must Read
The Salty Seas and Earthy Delights
One of the most common places to find alkali metals is in seawater. Yep, that salty taste? Thank sodium, an alkali metal, bonding with chlorine to form sodium chloride – good ol' table salt!
These elements are also found in various minerals within the Earth’s crust. Lithium, for example, can be found in rocks like spodumene and petalite. Imagine these reactive fellas just chilling underground, waiting for a chance to mingle.
Potassium, another important alkali metal, is abundant in minerals like sylvite and carnallite. These minerals contribute to soil fertility, making potassium essential for plant growth. Who knew our leafy friends relied on these energetic elements?
A Deep Dive into the Periodic Neighborhood
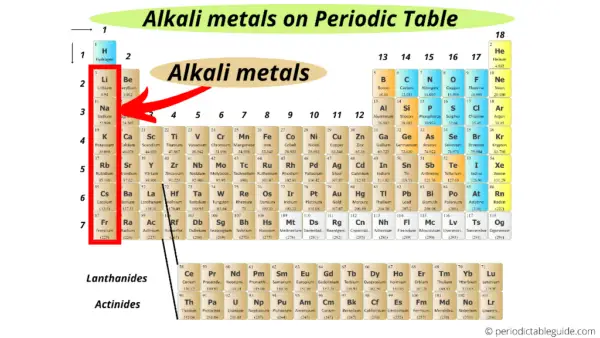
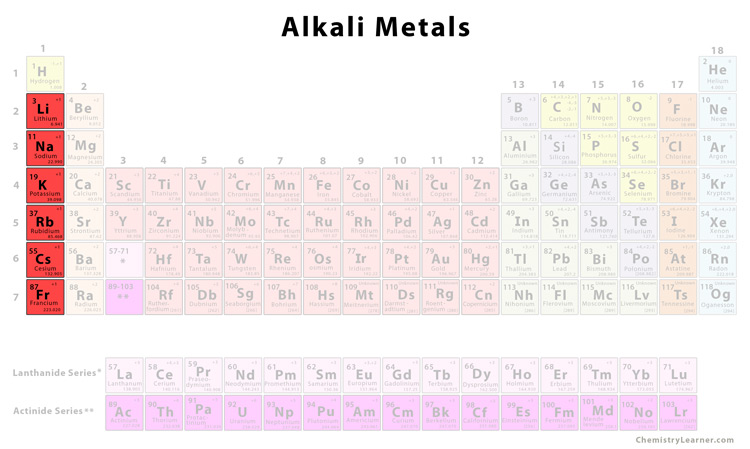
Alright, let's get a bit more specific about their location on the periodic table. These guys all live in Group 1, the very first column on the left. You could say they're the trendsetters of the element community.
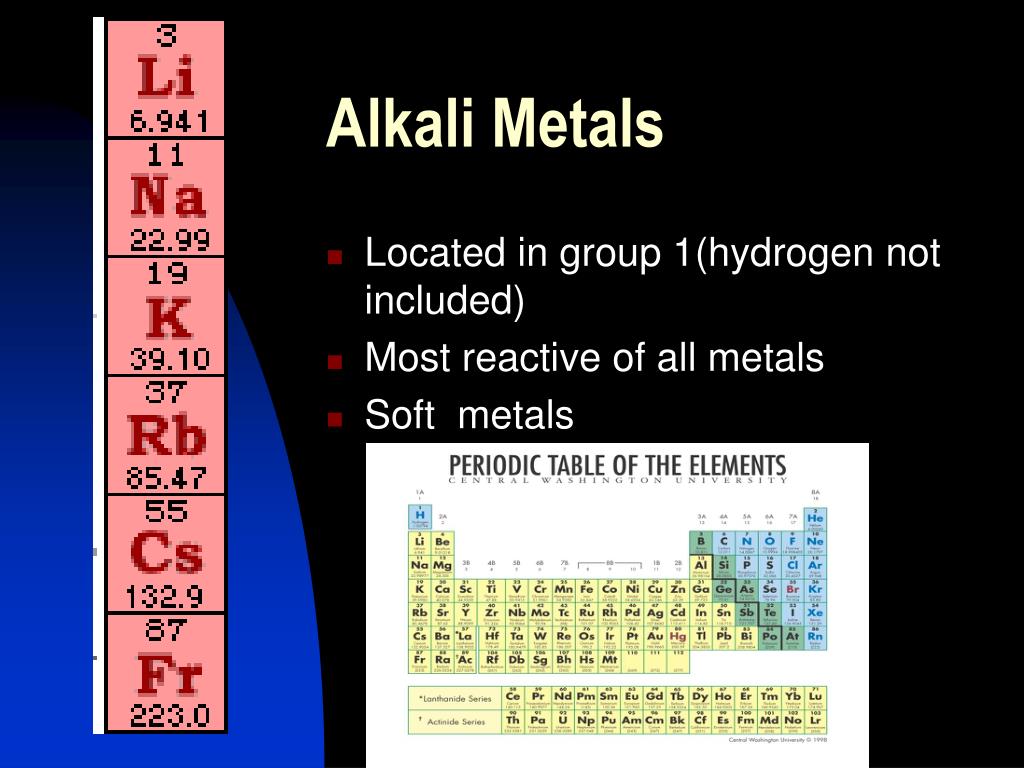
This group includes lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). Each has unique properties, but they all share a common characteristic: a single valence electron, which makes them incredibly reactive.
Think of it like having one lonely sock – they're desperate to find a pair! This eagerness to bond makes them incredibly useful in various applications.
Unlocking the Alkali Secrets: Uses in Everyday Life
Beyond their reactivity, these elements play essential roles in our daily lives. From keeping us hydrated to powering our devices, alkali metals are secretly working behind the scenes.
Sodium: The Body's Best Friend
We've already mentioned table salt, but sodium does so much more. It's vital for nerve function, muscle contractions, and maintaining fluid balance in our bodies.
Just remember to keep your intake in check – too much sodium can be a problem. Moderation is key, even for the most energetic elements.
Potassium: Powering Our Plants and Bodies
Potassium is another electrolyte that's crucial for our health. It helps regulate blood pressure, supports nerve function, and plays a role in muscle contraction.
It's also essential for plant growth. Fertilizers often contain potassium compounds to help plants thrive. So, thank potassium for those delicious fruits and veggies on your plate!
Lithium: A Mood-Boosting Marvel
Lithium has a fascinating role in mental health. Lithium carbonate is used as a mood stabilizer in treating bipolar disorder.

The precise mechanism isn't fully understood, but it's believed to affect neurotransmitter activity in the brain. It's like giving the brain a gentle, supportive hug.
Rubidium and Cesium: Timekeepers of the Atomic World
Rubidium and cesium are used in atomic clocks. These clocks are incredibly precise, losing or gaining only a second over millions of years!
Cesium atomic clocks are so accurate, that they are used to define the second! Talk about being punctual.
Francium: The Elusive Enigma
Francium is a rare and radioactive element. It's so unstable that it quickly decays into other elements. Because of its extreme radioactivity, it is the rarest element on earth.
Due to its scarcity, francium doesn't have many practical applications. It remains a subject of scientific curiosity, a fleeting glimpse into the world of highly unstable elements.
The Alkali Metal Legacy: A Colorful Conclusion
So, where are the alkali metals located? Everywhere! From the oceans to the Earth's crust, and even inside our bodies.
They are the active social butterflies of the periodic table. They’re always eager to react and form compounds.
These elements remind us that even the most reactive personalities can play vital roles in our world. Whether it's keeping us healthy, powering our devices, or simply adding a dash of salt to our meals, alkali metals are indispensable. They are a testament to the power and the magic hidden within the elements.
