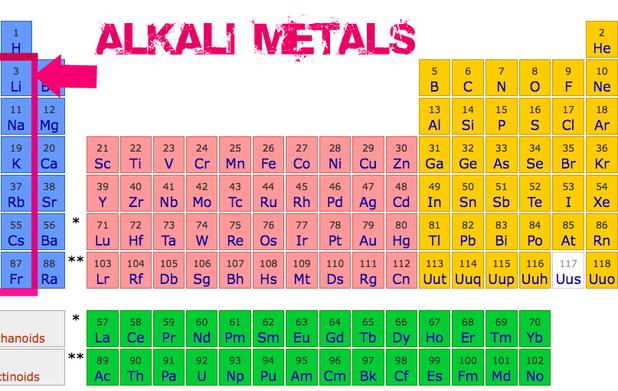
Where Are The Alkali Metals In The Periodic Table
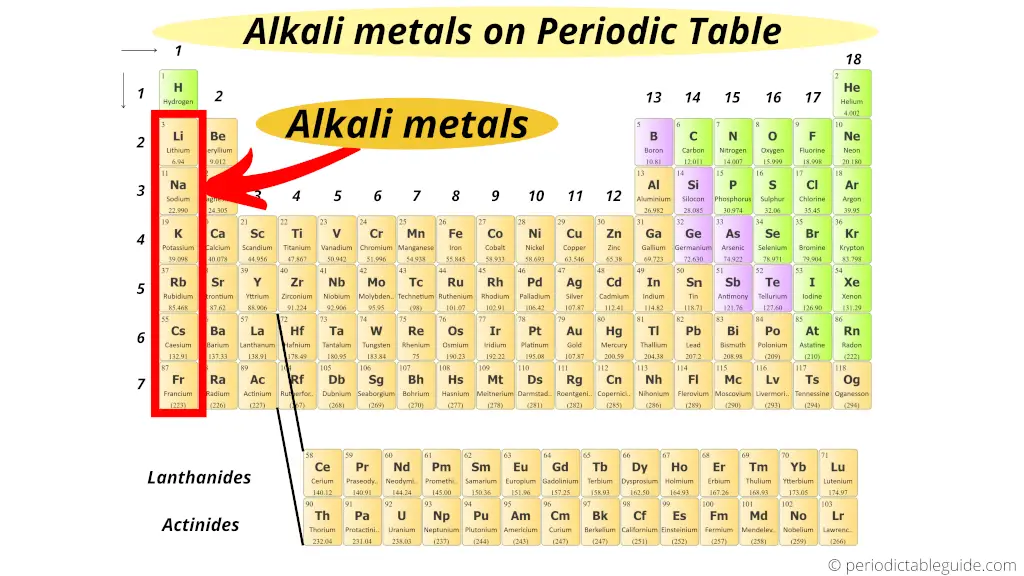
Hey there, chemistry chum! Ever wonder where to find those super-reactive, fizz-in-water elements known as the alkali metals? Well, grab your periodic table (or just picture one in your mind – I won't judge!) and let's go on a treasure hunt!
Hunting for the Hooligans of Group 1
Seriously, if the periodic table were a high school, the alkali metals would be the super-active kids always getting into trouble (but in a fun, science-y way, of course!). They're located in Group 1, that very first column on the left-hand side of the periodic table. Easy peasy, right?
Think of it like this: Group 1 is where the cool kids hang out. Okay, maybe "cool" isn't the right word. More like... explosively reactive kids. Yeah, that's more like it.
Must Read
The Usual Suspects (and Their Shiny Personalities)
So, who are these rascals? Let's run through the lineup:
- Lithium (Li): The lightest of the bunch. You'll find it in batteries, keeping your phones and laptops buzzing. So, next time your phone's at 1%, thank Lithium!
- Sodium (Na): You know, like in sodium chloride… aka, table salt! It's essential for life (though maybe don’t lick the pure sodium – that’s a bad idea).
- Potassium (K): Another essential nutrient! You get it from bananas (the go-to snack for potassium power!), and it's vital for nerve function. Keep that potassium coming to avoid any grumpy potassium deficiency moods!
- Rubidium (Rb): Getting into slightly less common territory now. Rubidium is used in some specialized applications like atomic clocks! So next time you're precisely on time, think rubidium!
- Caesium (Cs): Even rarer than rubidium. Also used in very precise atomic clocks, and it’s even cool enough to be used in some ion propulsion systems! Talk about out of this world!
- Francium (Fr): The rarest and most unstable of the alkali metals. It's so radioactive that it's practically impossible to study in large quantities. A true elusive element.
Notice anything about their names? They all end in "-ium"! That's a handy way to remember them. Except, you know, for sodium and potassium... gotta keep you on your toes!
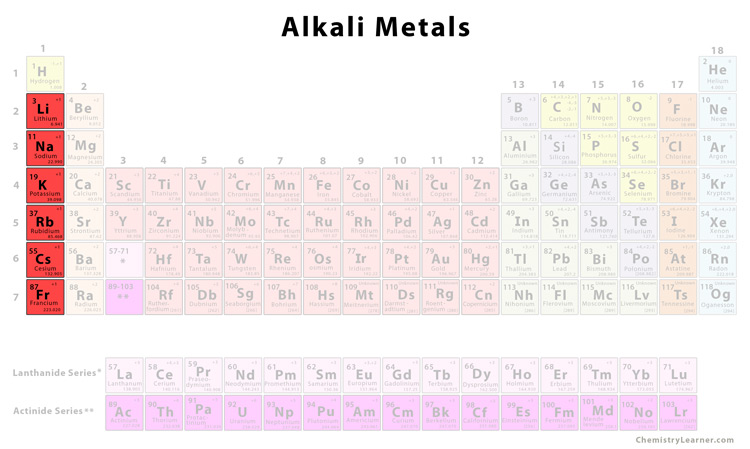
Why Are They So… Reactive?
Alright, so why are these guys so eager to react with everything? It all boils down to their electron configuration. Each alkali metal has only one valence electron – that's a single electron in its outermost shell. They're desperate to get rid of it to achieve a stable, full outer shell (like the noble gases, their neighbors to the right on the periodic table!).
Think of it like having that one annoying piece of popcorn stuck in your teeth. You just have to get rid of it, right? The alkali metals feel the same way about that lone electron. They're willing to give it away to pretty much anyone who asks nicely (or not so nicely!). And that, my friend, is what makes them so reactive.
A Word of Caution (Don't Try This at Home!)
Just a friendly reminder: don't go grabbing chunks of pure sodium and throwing them in water. Seriously. You'll get a fiery explosion and probably scare your cat. Leave the alkali metal experiments to the professionals (or at least watch them on YouTube!).
These elements are highly reactive, meaning they react vigorously (and sometimes violently) with water, air, and other substances. Store them under oil, not in your pocket!

So, Where Are They Again?
Okay, quick recap: The alkali metals live in Group 1 of the periodic table, the far left column. They're the super-reactive elements with one valence electron just itching to be given away. They're essential for all sorts of things, from powering your phone to keeping your heart beating. Pretty cool, huh?
Now go forth and impress your friends with your newfound alkali metal knowledge! You're practically a chemistry rockstar now!
Chemistry can seem daunting but remember every time you use the elements, like the Alkali metals, you are interacting with the very building blocks of the universe. And knowing where to find them and a little about them? That's a superpower in itself. So keep learning, keep exploring, and keep that brilliant mind of yours shining bright. You're awesome!