Where Are Metalloids On The Periodic Table
Hey there, fellow science enthusiasts! Ever stumbled upon the term "metalloid" and wondered what it's all about? Or, more importantly, where exactly these mysterious elements hang out on the periodic table? Well, you're in the right place! Let's dive into this intriguing topic, shall we?
The Periodic Table: Our Map of the Elements
First things first, the periodic table. Think of it as the ultimate cheat sheet to understanding all the building blocks of, well, everything! It neatly organizes elements based on their properties. But where do metalloids fit into this grand scheme?
Imagine the periodic table as a big neighborhood. You've got your super-outgoing metals on one side, always eager to bond with everyone. Then you have the introverted nonmetals on the other side, a bit more selective about their interactions. And right in the middle, acting as the friendly mediators, are our metalloids!
Must Read
So, Where Exactly Are They?
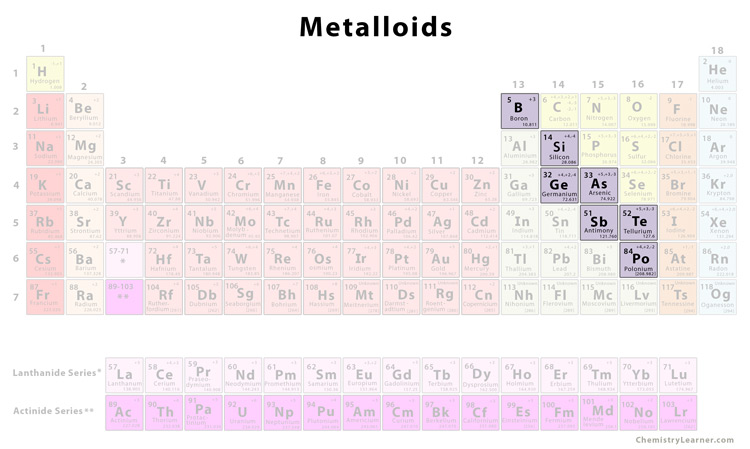
Okay, let's get specific. Metalloids, also known as semimetals, aren't neatly grouped into one specific block like the metals or nonmetals. They’re kind of sprinkled along a diagonal line, almost like a staircase, separating the metals from the nonmetals. Cool, right?
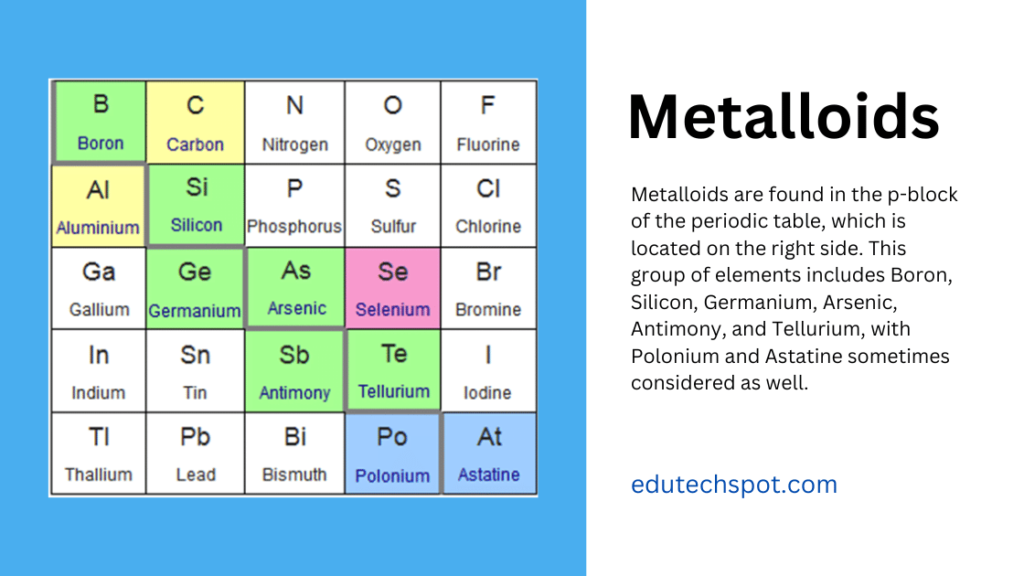
You'll typically find them clustered around elements like boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), and polonium (Po). That's quite a mouthful, I know! Sometimes, astatine (At) is also considered a metalloid, but it's a bit more debatable due to its radioactive nature and limited study.
Think of it like this: metals are the loud partygoers, nonmetals are the quiet bookworms, and metalloids? They're the ones hanging out in the kitchen, having interesting conversations with both groups. They're bridging the gap!
Why Are Metalloids So Interesting?
Now, why should we even care about these halfway-there elements? The real magic of metalloids lies in their unique properties. They can act like metals under certain conditions and like nonmetals under others. It's like they have a secret identity!
This chameleon-like behavior makes them incredibly useful in all sorts of technologies. The most famous example? Silicon. It's the bedrock of the entire semiconductor industry! Without silicon, your computers, smartphones, and pretty much any modern electronic device wouldn't exist. Crazy, right?

Imagine trying to build a super-fast computer out of just metals or just nonmetals. It just wouldn't work! Metals are too conductive, meaning they let electricity flow too freely. Nonmetals, on the other hand, are insulators, meaning they don't let electricity flow at all. But metalloids? They're the Goldilocks of elements – just the right amount of conductivity.
Germanium is another key player, also used in semiconductors, although less common than silicon these days. Arsenic and antimony are used in alloys to increase hardness and improve corrosion resistance. Tellurium is used in solar panels to improve their efficiency. These elements might not be household names, but they're quietly powering our world!
It's All About the Electrons!
So, what's the secret behind their dual nature? It all boils down to their electron configuration. Electrons are the tiny particles that orbit the nucleus of an atom, and their arrangement determines how an element will interact with other elements.

Metalloids have electron configurations that are somewhere between those of metals and nonmetals. This means they can sometimes lose electrons like metals, and sometimes gain electrons like nonmetals. It's all about context!
Think of it like this: metals are generous and like to give away electrons. Nonmetals are greedy and like to take electrons. Metalloids are more like negotiators – they're willing to share or trade electrons depending on the situation.
The Fuzzy Boundaries
It’s important to note that the classification of elements as metalloids can sometimes be a bit fuzzy. There’s not always a strict consensus on which elements exactly qualify. It often depends on the specific properties being considered and the particular scientific context.
But that's part of what makes science so exciting, right? It's not always about black and white answers. There's always room for debate and further exploration.
Metalloids: The Unsung Heroes
So, next time you're glancing at the periodic table, remember the metalloids! They might not be as flashy as the alkali metals or as reactive as the halogens, but they play a crucial role in our modern world.
They are the unsung heroes of the element world, quietly enabling the technologies that power our lives. Who knew that these seemingly ordinary elements could be so extraordinary? Keep exploring and keep questioning, you never know what amazing discoveries await!
