Where Are Metalloids Located On The Periodic Table

Okay, let's talk metalloids. Now, I know what you're thinking: "Metalloids? Sounds like something out of a sci-fi movie!" But trust me, they're way more common than laser swords and teleportation devices. Think of them as the "Switzerland" of the element world – neutral, adaptable, and chill.
So, where do we find these neutral buddies on the periodic table? It's like trying to find the best coffee shop in a new city. You know it's somewhere around, but you need a little help.
The Staircase to Metalloid-dom
Imagine the periodic table as a massive, slightly intimidating, rectangular building. All the elements are residents living in perfectly organized apartments. The metalloids live on a special staircase that separates the super-shiny, always-eager-to-give-away-electrons metals from the sometimes-grumpy, always-eager-to-grab-electrons nonmetals. They’re the folks who can be either, depending on who they're hanging out with.
Must Read
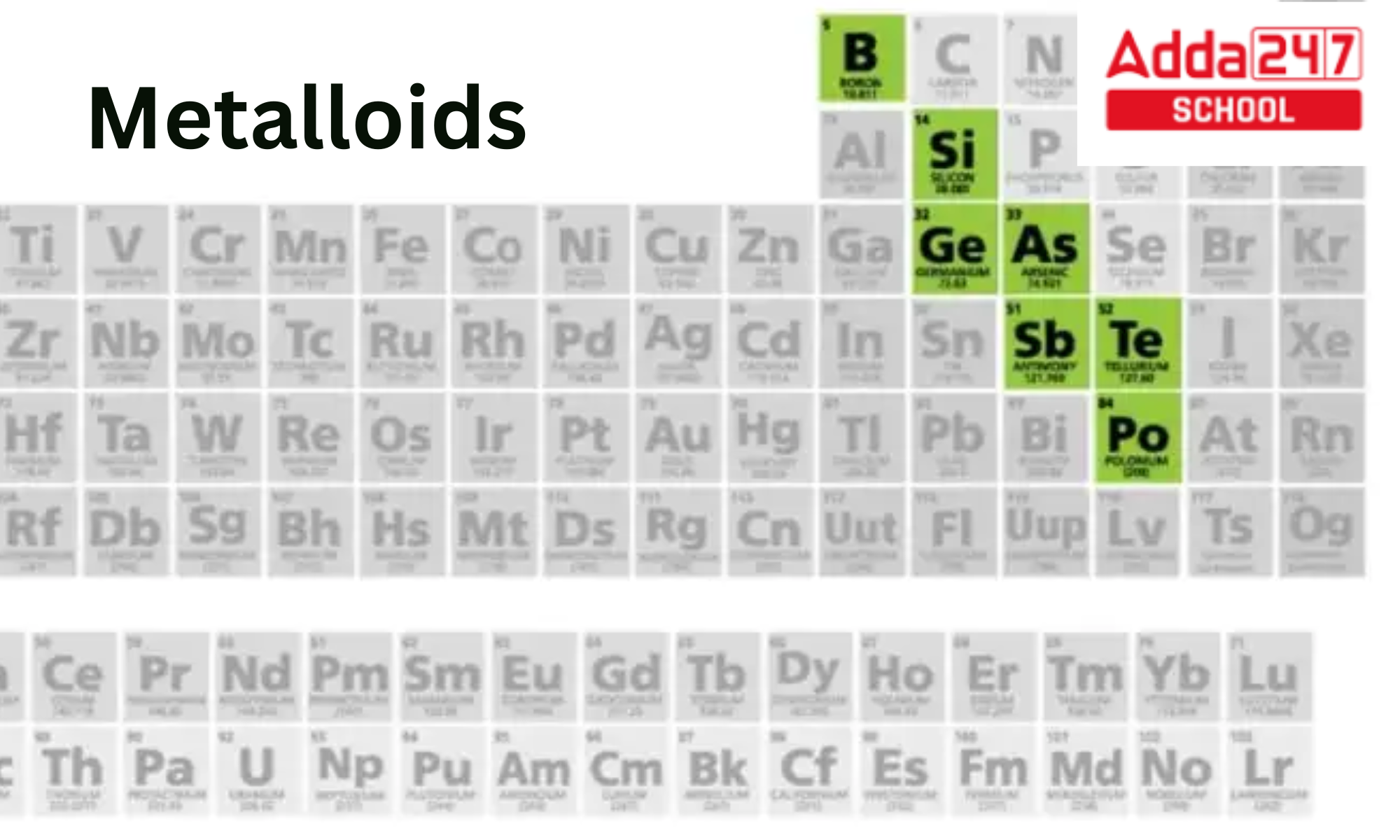
That staircase? It's formed by a diagonal line, starting around Boron (B) and descending down towards Astatine (At). This is your metalloid real estate!
Think of it like this: metals are the extroverted party animals, nonmetals are the introverted bookworms, and metalloids are the ambiverts who can mingle at the party and enjoy a quiet night with a good book. They're flexible! And their location is key to that flexibility.
Meet the Neighbors
Let's name a few metalloid neighbors, shall we?
Boron (B): First on the staircase! Boron compounds are used in things like borax (that cleaning stuff your grandma always used) and some types of glass.
Silicon (Si): Ah, silicon! The king of semiconductors. This guy is in practically every computer chip you own. Your phone? Silicon. Your laptop? Silicon. That fancy toaster oven with the digital display? You guessed it – silicon!

Germanium (Ge): Another semiconductor star! You might not run into germanium every day, but it's still a crucial part of many electronic devices.
Arsenic (As): Okay, arsenic has a bit of a shady past (think old detective novels). But it's also used in some alloys and semiconductors. Don't worry, you're not likely to encounter dangerous levels of it in your daily life... unless you're starring in a murder mystery.
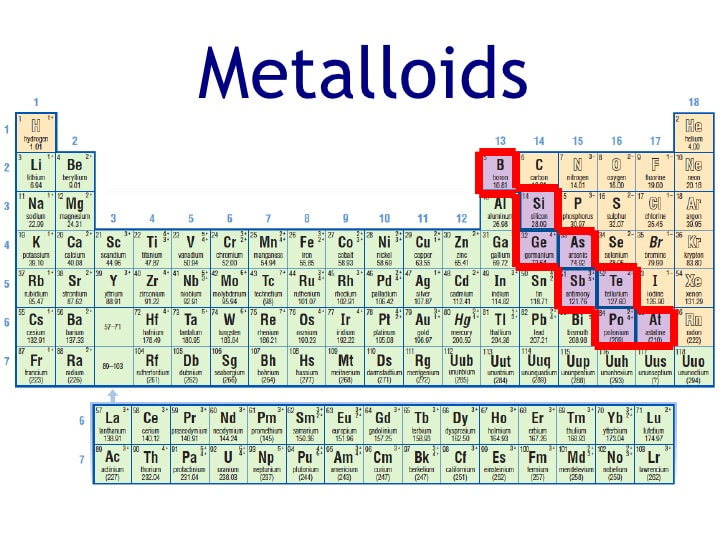
Antimony (Sb): Antimony is often used to make things fire-retardant. So, next time you see that "fire-resistant" label, thank antimony!
Tellurium (Te): Tellurium is a rare one, often used in solar panels and alloys. It’s a bit of a specialist!
Polonium (Po): Metalloid or not? That is the question. Polonium is a radioactive element that can act like a metalloid, and is located near the "bottom" of the staircase. Let's just say it's complicated!

Why Their Location Matters
So, why are these elements located where they are? It all boils down to their electronic properties. They have properties that are intermediate between metals and nonmetals, allowing them to conduct electricity under certain conditions. This is why they're so important in electronics. They're not quite conductors (like metals), and not quite insulators (like nonmetals), making them perfect for controlling the flow of electricity.
Think of it like having a dimmer switch for your lights. Metals are like having the light switch permanently on, and nonmetals are like having it permanently off. Metalloids are the dimmer switch, allowing you to adjust the brightness as needed.
So, next time you're using your phone, or browsing the internet on your computer, remember the metalloids. They're the unsung heroes of the electronic age, chillin' on their staircase on the periodic table, making our digital lives possible. Now, if you'll excuse me, I need to go thank the metalloids by using a device powered by, well, them.
