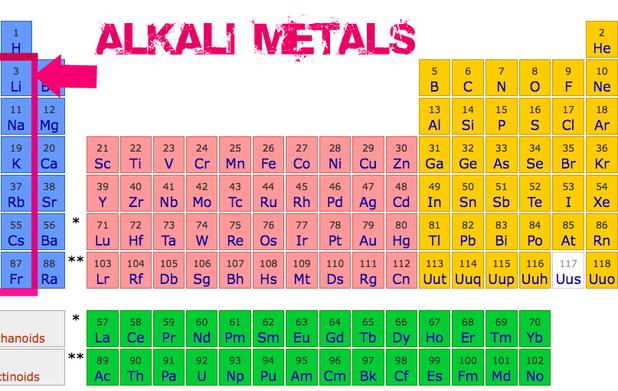
Where Are Alkali Metals On Periodic Table

Hey there, fellow knowledge seekers! Ever wondered about those enigmatic elements chilling out on the left side of the Periodic Table? We're talking about the Alkali Metals! They’re like the cool kids in chemistry class – reactive, shiny (at least when you first cut them!), and always ready to make a statement.
Location, Location, Location: The Alkali's Neighborhood
So, where exactly can you find these metallic marvels? Look all the way to the leftmost column (Group 1) of the Periodic Table, excluding hydrogen (that rebel does its own thing). They're lined up nice and neatly, starting with Lithium (Li) at the top, then Sodium (Na), Potassium (K), Rubidium (Rb), Cesium (Cs), and finally Francium (Fr). Think of them as the opening act for all the other elements.
Fun Fact: Francium is incredibly rare and radioactive. Scientists estimate that only about an ounce of it exists on Earth at any given time! Talk about exclusive.
Must Read
Meet the Family: An Alkali Metal Lineup
Let's get acquainted with a few of the key players:
- Lithium (Li): Beyond batteries, lithium compounds are used to treat mood disorders.
- Sodium (Na): Ever sprinkle salt on your fries? That's sodium chloride! Sodium is essential for nerve function and fluid balance.
- Potassium (K): Bananas are famous for their potassium content, which is vital for heart health. Potassium also helps regulate blood pressure.
- Rubidium (Rb) & Cesium (Cs): These guys are a bit more niche, but they're used in atomic clocks, which are incredibly precise timekeepers.
Pro Tip: Remember the order? Try this mnemonic: "Little Ninjas Kick Red Cats." Silly, but effective!
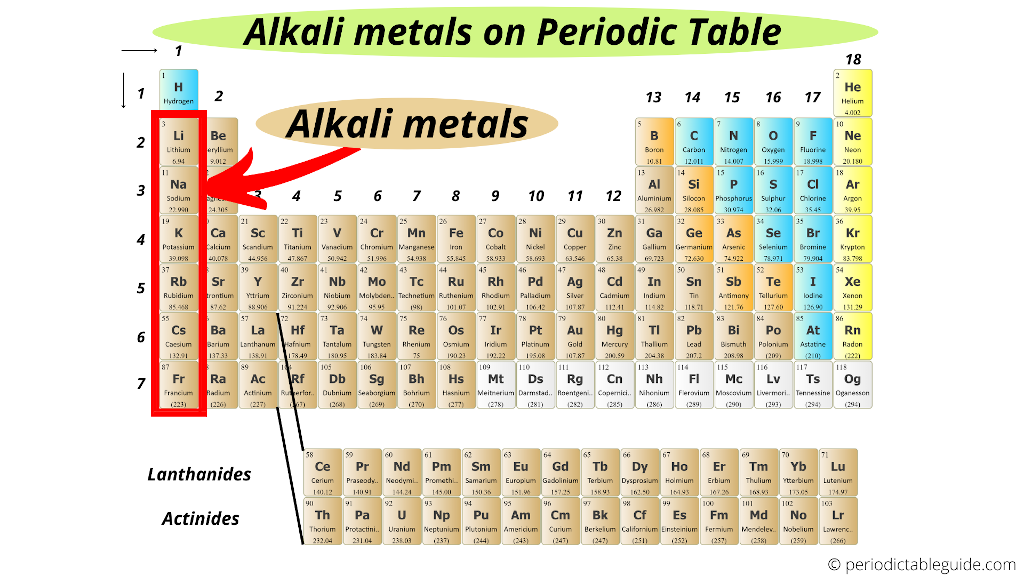
Why So Reactive? The Alkali Personality
What makes these metals so special, so… reactive? It all comes down to their electron configuration. They each have just one valence electron – that lone electron in their outermost shell. They're desperate to get rid of it to achieve a stable, full outer shell. This eagerness to donate their electron makes them highly reactive with other elements, especially halogens (Group 17 on the Periodic Table – the Alkali Metals' natural frenemies!).
Think of it like this: Imagine you have only one slice of pizza left, and you're on a diet. You'd be pretty quick to give it away, right? Alkali metals are the same with their valence electrons.
Alkali Metals in Everyday Life: More Than You Think
You might not realize it, but alkali metals play a crucial role in your daily life. Here are just a few examples:
- Table Salt (NaCl): As mentioned earlier, sodium chloride is essential for seasoning food and maintaining bodily functions.
- Batteries: Lithium-ion batteries power your smartphones, laptops, and electric vehicles.
- Soap: Many soaps are made using sodium or potassium hydroxide (lye).
- Fertilizers: Potassium is a key ingredient in fertilizers, helping plants grow strong and healthy.
Cultural Connection: Remember that scene in "Breaking Bad" where Walter White uses lithium aluminum hydride? Okay, don't try that at home. But it shows that even in pop culture, these elements can pop up in unexpected places!
Safety First! Handling Alkali Metals
A word of caution: alkali metals are not something you should handle casually. They react violently with water, producing hydrogen gas and heat. This can result in explosions and burns. In the lab, they're typically stored under oil to prevent them from reacting with air or moisture.
Practical Tip: Leave the alkali metal experiments to the professionals! Appreciate them from afar (or in a well-controlled laboratory setting).
Wrapping Up: The Alkali Impact
So, there you have it – a crash course on alkali metals! From their prime location on the Periodic Table to their essential roles in our daily lives, these elements are far more important than you might have thought. They remind us that even the most seemingly basic elements can have a profound impact on the world around us.
Next time you sprinkle salt on your food or use your phone, take a moment to appreciate the amazing chemistry happening at the atomic level. It's a reminder that everything, even the smallest things, are interconnected. Now, go forth and impress your friends with your newfound alkali metal knowledge!