What Makes Each Amino Acid Unique
Alright, gather 'round, folks! Let's talk about amino acids – the building blocks of life! Now, I know what you're thinking: "Amino acids? Sounds boring!" But trust me, these little guys are way more exciting than they sound. Imagine them as tiny LEGO bricks, each with its own quirky personality, all coming together to build, well, you. And me. And your dog. Pretty much everything alive.
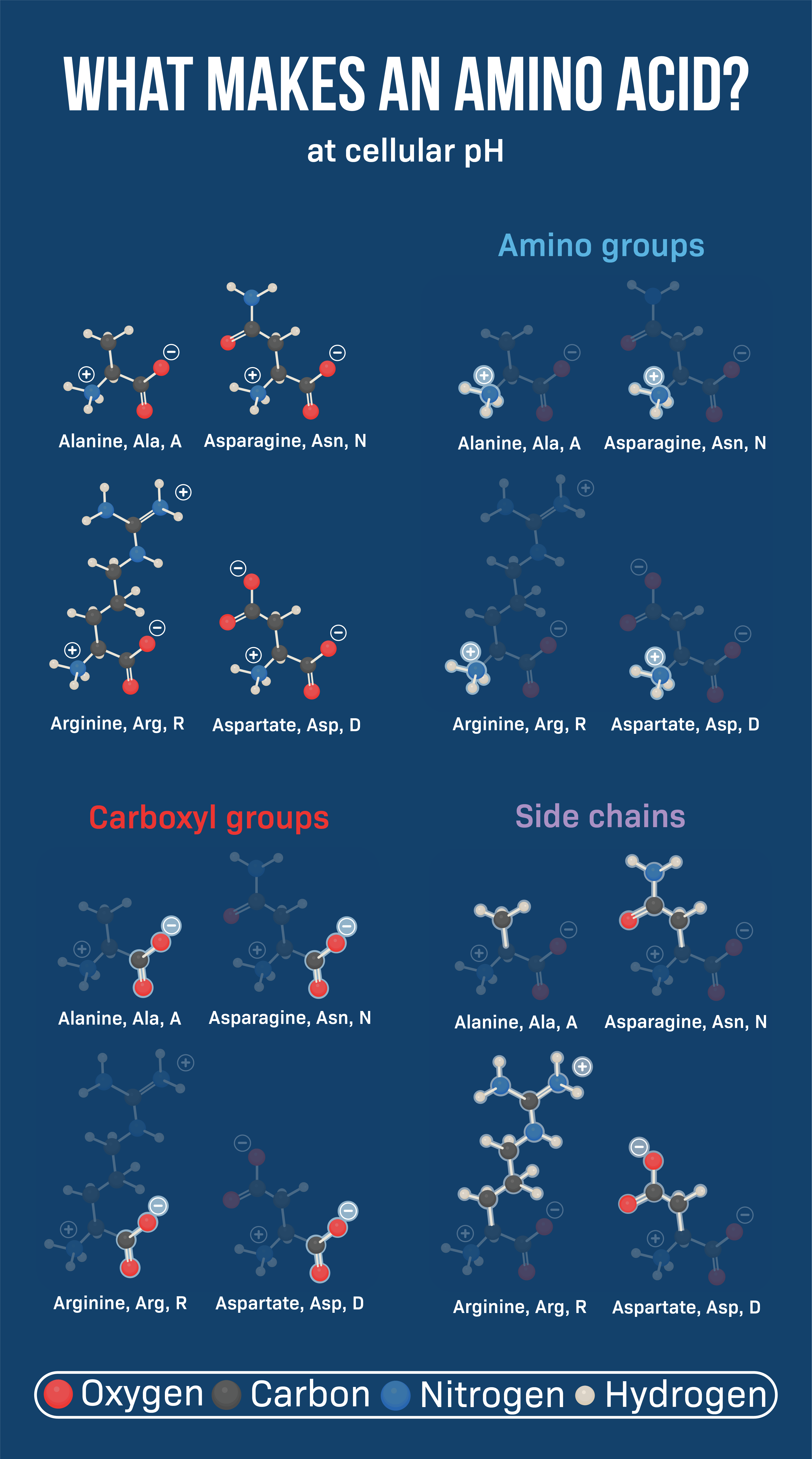
So, what makes each amino acid unique? It all boils down to their R-group, also known as the side chain. Think of it like this: every amino acid has the same basic body – a central carbon atom (the cool kid in the middle), an amino group (the friendly, outgoing one), a carboxyl group (the slightly grumpy, acidic one), and a hydrogen atom (the quiet observer). But the R-group? That's where things get wild. That's their personal style, their defining characteristic, their reason for existing (besides, you know, building proteins).
The Gang of Twenty (ish)
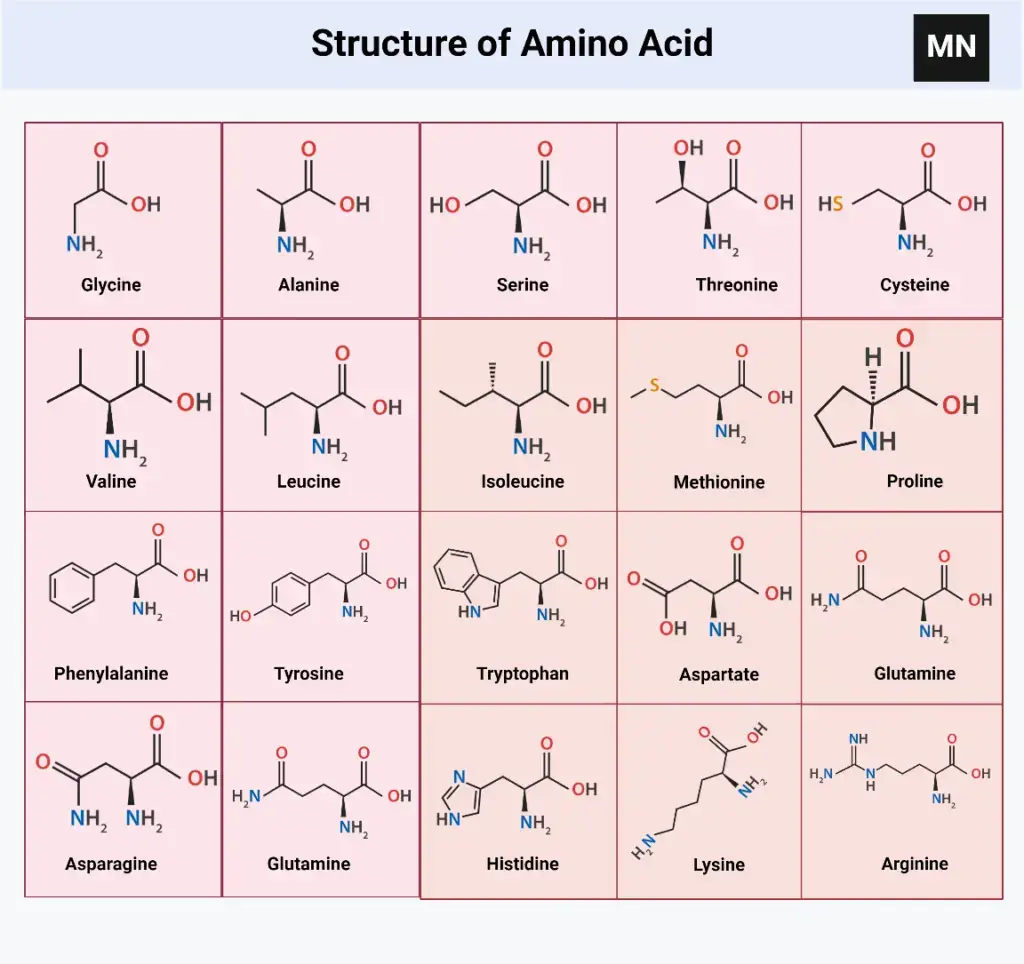
There are about 20 standard amino acids that your body uses to construct proteins. Think of them as the main cast in the protein-building movie that is your life. Each has a different R-group, giving it unique properties. Let's meet a few of the more memorable ones.
Must Read
First up, we have Glycine, the simplest of the bunch. Glycine is basically the minimalist of the amino acid world. Its R-group is just a single hydrogen atom. Seriously, that’s it. It's so small, it can squeeze into places other amino acids can't. It's like the yoga instructor of the protein world, all flexible and bendy.
Then there's Alanine. Alanine's R-group is a methyl group (CH3), which is like a single carbon with three hydrogens attached. It's a little bit bigger than Glycine, a little bit more... assertive. Alanine is pretty content just chilling out and not causing too much trouble.

Next, let's talk about the aromatic ring amino acids, which include Phenylalanine, Tyrosine, and Tryptophan. These guys are like the rock stars of the amino acid world. They have these big, bulky, ring-shaped R-groups that can absorb UV light. They are responsible for much of a protein’s UV absorbance properties. I’m sure you feel much better knowing that.
Cysteine is another fascinating amino acid because it contains sulfur. Two cysteines can bond together to form a disulfide bridge, adding extra stability to a protein. Think of it like protein handcuffs! It's also the amino acid most responsible for that lovely rotten egg smell. Yum!

Polar vs. Nonpolar: A Clash of Titans
Amino acids are generally categorized as either polar or nonpolar, based on their R-group. This difference in polarity has a huge impact on how proteins fold and function.
Nonpolar amino acids are like introverts. They don't like water (hydrophobic) and prefer to hang out in the interior of proteins, away from the watery environment of the cell. Think of them as the hermits of the amino acid world.
Polar amino acids, on the other hand, are like extroverts. They love water (hydrophilic) and are happy to interact with the surrounding environment. They often hang out on the surface of proteins, helping them interact with other molecules. They're the social butterflies of the amino acid world.
Acidic and Basic Amino Acids: The Extremes
Some amino acids are considered acidic or basic, depending on whether their R-group can donate or accept a proton (H+), respectively. These charged amino acids play important roles in enzyme catalysis and protein-protein interactions.
Aspartic acid and Glutamic acid are the acidic amino acids. They're like the sourpusses of the group, always donating protons and acting negatively charged at physiological pH.

Lysine, Arginine, and Histidine are the basic amino acids. They're like the cheerful optimists, always accepting protons and acting positively charged (or at least neutral in the case of Histidine, which is a bit of a fence-sitter depending on the pH of its surroundings.)
So, there you have it! A whirlwind tour of the amino acid world. Each amino acid is unique, with its own distinct R-group that gives it specific properties and contributes to the overall structure and function of proteins. Remember, next time you're eating a protein-rich meal, you're not just consuming generic "protein." You're consuming a carefully curated collection of amino acids, each playing its part in the grand symphony of life.
And if anyone ever tells you amino acids are boring, just tell them to come talk to me. I’ll set them straight (after I’ve had a coffee, of course).
