What Is Thermal Energy Simple Definition

Okay, so picture this: you're making toast. Maybe you’re a fancy avocado toast kinda person, or maybe you’re a plain-butter-is-fine-thank-you type (no judgment!). Either way, you pop that bread into the toaster, and after a bit, it comes out… golden brown and magically crispy. But what really happened in that toaster besides the obvious browning? Well, that, my friends, is where thermal energy comes in. And no, it's not magic (although toasted bread IS pretty magical!).
Think about it. The toaster's heating elements got hot, right? That heat transferred to the bread, changing its texture and color. That transfer of heat? Yep, that’s thermal energy at work. So, what is thermal energy exactly?
The Super Simple, No-Brainer Definition of Thermal Energy
Alright, let’s ditch the physics textbook jargon. Thermal energy is simply the energy an object has due to the movement of its atoms or molecules. Everything is made of these tiny little particles, and they're always jiggling, vibrating, or zooming around like tiny, caffeinated hamsters.
Must Read
The faster these particles move, the more thermal energy the object has, and the hotter it feels. The opposite is also true! The slower they move, the less thermal energy and the colder it becomes. Makes sense, right?
Think of it like this: a bunch of excited kids running around a playground (high thermal energy = hot!) versus a group of sleepy kittens napping in a sunbeam (low thermal energy = cold!). I know which scenario I prefer... especially on a Monday morning!

But Wait, There’s More! (Because Science is Never Really Simple, Is It?)
Okay, so while the "jiggling particles" explanation is a good starting point, it’s important to remember that thermal energy is related to temperature and heat, but it’s not exactly the same thing. (I know, I know... it's like a Russian doll of scientific terms!)
Temperature is a measure of the average kinetic energy of the particles in an object. It tells you how hot or cold something is on a scale (like Celsius or Fahrenheit). Thermal energy, on the other hand, is the total energy of all those moving particles. (So, it depends on both temperature and how much "stuff" there is!).
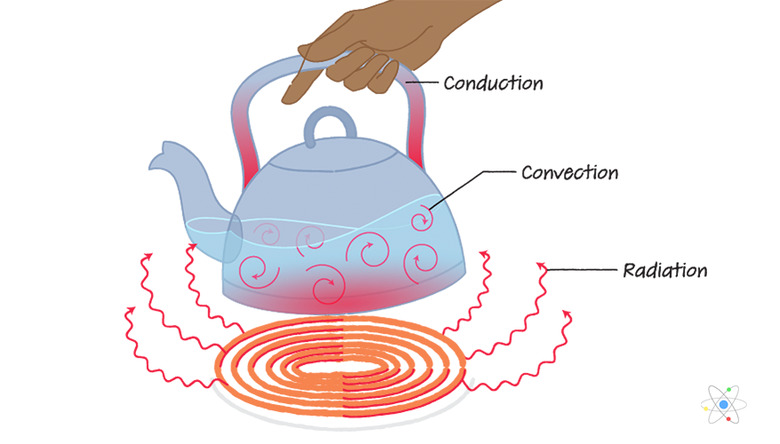
Heat is the transfer of thermal energy from one object to another. Think back to that toast. The heat from the toaster’s elements transferred thermal energy to the bread. The bread got hotter. The elements got slightly cooler. Everybody wins (except maybe the carbs!).
Side note: Heat always flows from hotter objects to colder objects. You’ll never see your ice cream warming up your hand (unless you’re living in some kind of upside-down world, which, let's be real, sometimes it feels like we are!).
Examples of Thermal Energy in Action (Because Everything Is Connected!)
Thermal energy is everywhere! Seriously, it's the unsung hero of the universe.
- The Sun: A giant ball of fiery thermal energy! Without it, we'd be frozen popsicles floating in space. (Not ideal.)
- Geothermal Vents: Undersea vents spewing out hot water heated by the Earth's core. Talk about a natural jacuzzi! (Except probably not safe to swim in.)
- Rubbing Your Hands Together: Ever noticed how your hands get warmer when you rub them together? That friction converts mechanical energy into thermal energy! Boom! You're a human heater.
- A Lightbulb: Ever touched a lightbulb after it's been on for a while? Ouch! That’s thermal energy as a byproduct of electricity flowing through the filament. (Please don’t touch hot lightbulbs, though!).
So, the next time you feel the warmth of the sun, brew a cup of hot tea, or just… exist, remember those tiny, buzzing particles doing their thing. It's all thanks to thermal energy – the energy of movement, the energy of heat, and the energy that makes our universe, well, warm and interesting. Now, if you'll excuse me, I'm going to go make some toast. (Because, let's be honest, all this talk about thermal energy has made me hungry!)