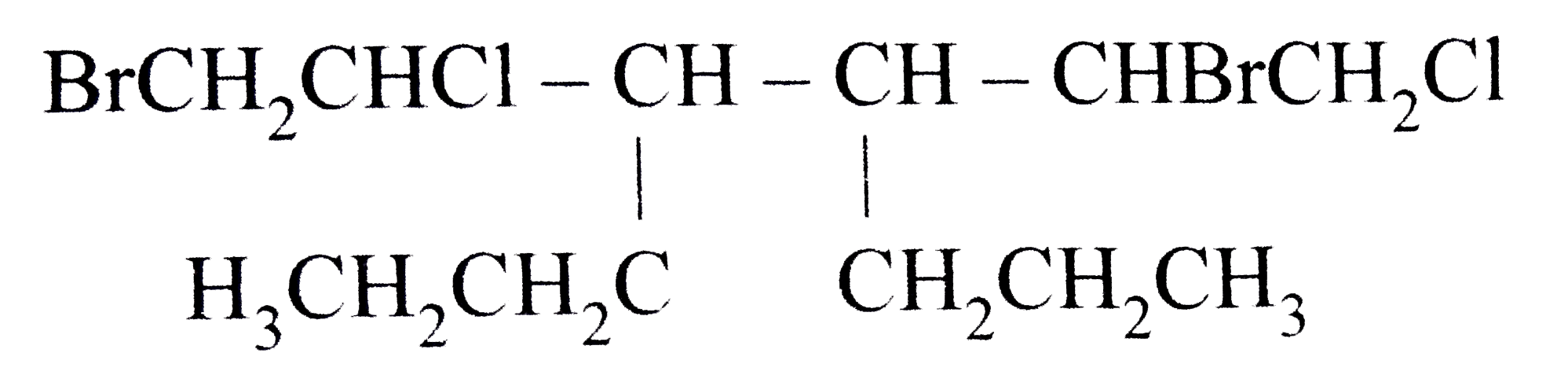
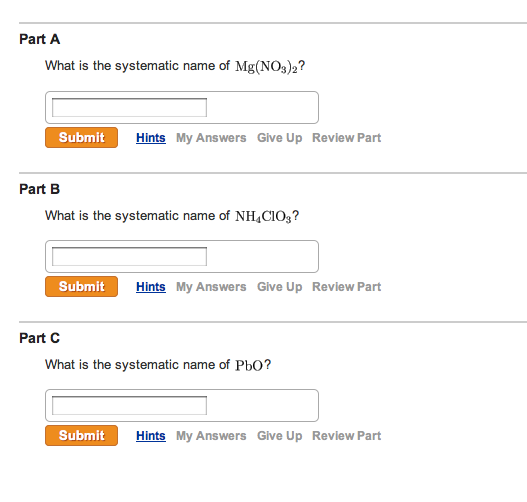
What Is The Systematic Name Of Nh4clo3
Okay, let's be honest. When was the last time you were at a party and someone casually dropped the phrase "Ammonium Chlorate" into conversation? Probably never! But before you dismiss it as some obscure chemistry term only understood by lab coat enthusiasts, let's explore why understanding the systematic name of NH4ClO3 – Ammonium Chlorate, in case you skipped ahead – can actually be surprisingly inspiring, especially if you're the creative type or just someone who enjoys learning new things.
You might be thinking, "What possible benefit could knowing this have for me as an artist or hobbyist?" Well, consider this: understanding the systematic naming of chemical compounds is like unlocking a secret code. It’s about understanding the structure behind something, the building blocks. And that understanding, even in a seemingly unrelated field like chemistry, can fuel creativity and enhance your problem-solving skills. Think of it as cross-training for your brain!
For artists, understanding chemical structures can inspire new forms and patterns. Imagine designing a sculpture based on the crystal structure of ammonium chlorate (don't actually try to make it, though! It’s an oxidizer and reacts strongly with organic materials and reducing agents, with a risk of fire and explosion, when heated). You could explore the visual representation of molecules, translating them into abstract paintings or digital art. The precise and ordered nature of the chemical world can be a powerful source of artistic inspiration.
Must Read
Hobbyists who enjoy things like model building or crafting can benefit too. Knowing the components of different materials helps you understand their properties and how they react. You might choose specific paints or adhesives based on their chemical compositions, leading to better results and greater longevity in your projects. It also allows for informed experimentation, understanding why certain combinations work or don’t.
Even for the casual learner, understanding the systematic name of NH4ClO3 unlocks a deeper appreciation for the world around us. It’s a tangible example of how scientists organize and classify information, making complex concepts accessible. Instead of just seeing a random chemical formula, you can break it down: "Ammonium" (NH4+) is the positive ion, and "Chlorate" (ClO3-) is the negative ion. Suddenly, it's not just a jumble of letters; it's a description of a molecule.

Tips for Trying This at Home (Safely!): Don't handle ammonium chlorate! Instead, focus on understanding how its name is derived. Research the rules for naming ionic compounds. Start with simpler examples like sodium chloride (table salt) or potassium iodide. Use online resources and interactive simulations to visualize the molecular structures. There are plenty of excellent chemistry websites and apps that make learning fun and engaging.
The beauty of understanding systematic names in chemistry isn't about memorizing lists. It's about the process of understanding. It's about developing critical thinking skills, appreciating the order in the universe, and finding unexpected connections between seemingly disparate fields. It’s about transforming a potentially dry subject into a source of inspiration and intellectual curiosity. And who knows? Maybe you will be the life of the party, casually dropping your newfound knowledge of chemical nomenclature. At the very least, you'll have a fascinating story to tell!