What Is The Shell Level For Thallium

Okay, let's be honest. "Shell level for thallium" probably isn't the hottest topic at your next dinner party. But stick with me! Understanding the atomic structure of elements, like thallium, is like knowing the secret ingredient that makes the entire universe tick. Think of it as cosmic detective work, piecing together the fundamental building blocks of everything around us. While you might not be using this knowledge to impress your friends directly, it empowers you to grasp the underlying principles behind countless technologies and natural phenomena, from the glow of your smartphone screen to the composition of distant stars.
So, what’s the big deal with electron shells, specifically for thallium? Understanding an element’s electron configuration, or shell level, unlocks its chemical behavior. It helps us predict how it will interact with other elements, whether it will form strong bonds or remain aloof. In short, it's the blueprint for how an element behaves in the chemical world. This knowledge is absolutely crucial for everything from designing new drugs to understanding environmental pollution.
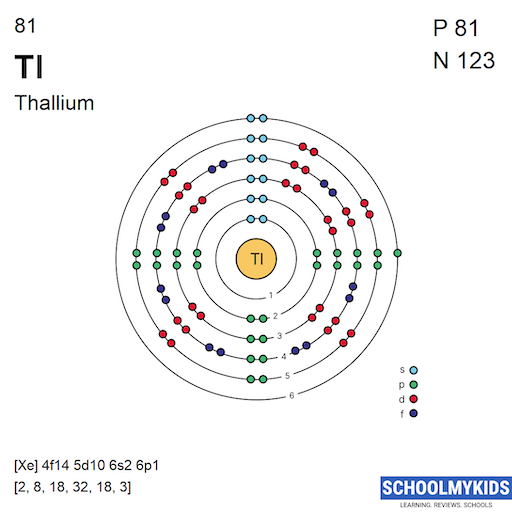
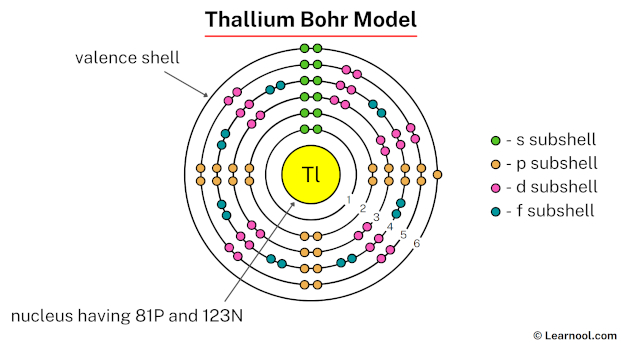
Thallium (Tl), residing at atomic number 81 on the periodic table, has a complex electron configuration. The electrons arrange themselves in specific shells around the nucleus. In thallium, these shells are filled in the following manner: 2 electrons in the first shell (K), 8 in the second (L), 18 in the third (M), 32 in the fourth (N), 18 in the fifth (O), and 3 in the sixth (P). This seemingly esoteric arrangement dictates thallium's properties. For example, it explains its tendency to form +1 and +3 oxidation states, which is essential to understanding its reactivity. This, in turn, explains how it behaves in different chemical reactions and how it can be used (or unfortunately, misused).
Must Read
While thallium itself isn't exactly a household staple, the principles governing its atomic structure are applied everywhere. Think about the development of new materials. Scientists use their understanding of electron configurations to design alloys with specific properties, making them stronger, lighter, or more resistant to corrosion. In medicine, this knowledge is vital for designing targeted drug delivery systems. Understanding how elements interact at the atomic level allows researchers to create molecules that bind to specific receptors in the body, maximizing therapeutic effects while minimizing side effects.
/Thallium-58b6010d5f9b5860464bac8b.jpg)
So, how can you appreciate the shell level of thallium more effectively (without becoming a quantum physicist)? Here are a few tips:
- Explore interactive periodic tables: Many online resources offer interactive periodic tables that allow you to visualize electron configurations and explore the properties of different elements.
- Watch documentaries about chemistry and materials science: These documentaries often explain complex concepts in an accessible and engaging way, showcasing the practical applications of atomic structure.
- Read popular science articles: Stay updated on new discoveries in chemistry and materials science. You'll be surprised how often these discoveries rely on understanding the electron configurations of elements like thallium.
- Remember the bigger picture: Don’t get bogged down in the details. Focus on the underlying principles and how they connect to real-world applications. The goal is to appreciate the elegance and interconnectedness of the universe, not to memorize a textbook.
Ultimately, understanding the shell level of thallium isn't just about memorizing numbers. It's about appreciating the fundamental principles that govern the universe and how these principles are harnessed to improve our lives. It’s about realizing that even seemingly obscure knowledge can unlock amazing possibilities.