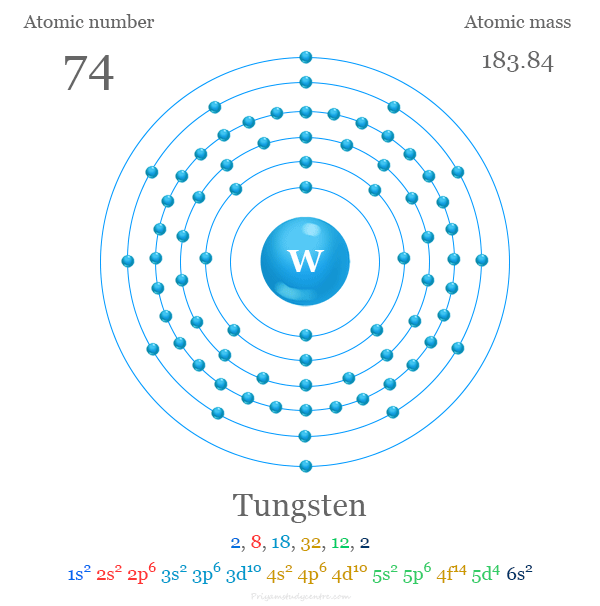
What Is The Atomic Mass Of Tungsten

Okay, so picture this: I'm at a trivia night, feeling all confident, right? The category is "Chemistry," and I’m thinking, “This is my time to shine!” Then the question pops up: "What is the atomic mass of Tungsten?" My brain promptly short-circuited. I could vaguely remember seeing 'W' on the periodic table, but the actual number? Gone. Poof. Vanished into the chemical ether. Let’s just say my team didn’t win that round. (Don't judge me! It happens to the best of us. 😉)
But that got me thinking: Tungsten, what is its deal? And more importantly, why did my brain betray me like that? So, I did what any self-respecting, slightly embarrassed trivia loser would do: I dove headfirst into researching the magnificent metal that is Tungsten.
What Exactly Is Atomic Mass Anyway?
Before we get to the specific number, let's do a quick refresher. Atomic mass isn't just some random number assigned to an element. It's the average mass of all the isotopes of an element, considering their relative abundance in nature. Think of it like this: you have a bunch of apples of slightly different sizes, and atomic mass is the average weight of all those apples. But the bigger apples count more towards the average! Get it? (If not, Google "isotopes" and "weighted average." Trust me, it’ll help.)
Must Read
It's measured in atomic mass units (amu), or Daltons (Da). Don't worry too much about the units; just know that they're incredibly small, perfect for measuring tiny atoms.
So, The Answer You've Been Waiting For: The Atomic Mass of Tungsten
Drumroll please... The atomic mass of Tungsten (W) is approximately 183.84 amu. Bam! There it is. Now, if I ever get that trivia question again, I’m READY. (And maybe you are too! Go you!)
You might see slightly different numbers depending on where you look. The value is actually based on a range because it's an average and can vary slightly depending on the source. But 183.84 is a very safe, generally accepted approximation.
Why Should I Care About the Atomic Mass of Tungsten?
Okay, valid question. You might be thinking, "Great, I know a random number. So what?" Well, understanding atomic mass is crucial in several scientific fields. For example:
- Chemistry: It's essential for calculating the amounts of substances needed for chemical reactions. You need to know how much stuff you're putting in, right?
- Material Science: Tungsten's high density and melting point make it super useful in various applications. Knowing its atomic mass helps scientists understand its properties and how it interacts with other materials. (Think light bulb filaments and radiation shielding!)
- Nuclear Physics: It plays a role in understanding nuclear reactions and the stability of isotopes. (This gets complicated fast, so maybe we'll save that for another day. 😉)
Basically, it's a fundamental piece of information for anyone working with Tungsten or related fields. It’s not just a trivia answer; it's a building block of scientific knowledge!

Fun Facts About Tungsten (Because Why Not?)
Let's make this a little more interesting, shall we?
- Tungsten has the highest melting point of all metals! That's why it's used in light bulb filaments. (Seriously, how cool is that?)
- Its symbol, 'W,' comes from the German name "Wolfram." (Mystery solved!)
- It's incredibly dense, making it useful in applications where you need a lot of weight in a small space. (Like, say, counterweights in aircraft.)
- Tungsten alloys are super strong and resistant to wear. (Think cutting tools and high-speed machinery.)
See? Tungsten is actually a pretty fascinating element! It's not just some random symbol on the periodic table.

In Conclusion: Go Forth and Impress People With Your Tungsten Knowledge!
So, there you have it! The atomic mass of Tungsten is approximately 183.84 amu. Now you're armed with this knowledge, you can confidently answer that trivia question (and maybe even win a prize!). More importantly, you have a better understanding of this important element and its role in the world around us.
And hey, if you ever forget the number again, just remember my trivia night disaster. We all have those moments. 😉 But now you know where to look!
Keep learning, stay curious, and maybe avoid chemistry trivia after a couple of beers. Just a thought.