What Is An Alkaline Earth Metal
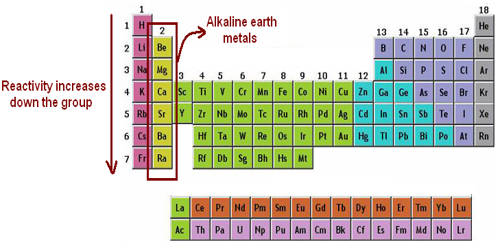
Ever wondered what makes your bones strong, or why fireworks explode in brilliant flashes of color? The secret often lies with a fascinating group of elements known as Alkaline Earth Metals. Don't let the fancy name scare you off; these are the unsung heroes of the periodic table, working tirelessly behind the scenes in everything from your morning coffee to the latest tech gadgets. Discovering them is like uncovering a hidden layer of magic in our everyday world, making science feel less like a textbook and more like a treasure hunt!
So, why dive into the world of alkaline earth metals? For beginners, understanding these elements provides a fantastic entry point into basic chemistry. You'll learn how common substances are formed and why certain elements behave the way they do, demystifying the building blocks of the universe right before your eyes. Families can turn this into a fun, educational adventure. Next time you're having milk, you can explain why calcium is so vital for growing bones. Or, during a fireworks display, point out how different metals create those spectacular reds and greens. For the curious hobbyist, knowing about these metals can deepen an appreciation for gardening (magnesium is key for healthy plants!), metallurgy (creating strong, lightweight alloys), or even just general scientific literacy, helping you understand product labels and news reports better.
Let's meet some of the stars of this group! Perhaps the most famous is Calcium (Ca). It’s not just in your dairy products; it's the primary component of your bones and teeth, gives eggshells their hardness, and is even used to make chalk and cement. Then there's Magnesium (Mg), an incredibly versatile metal. It’s at the heart of chlorophyll, giving plants their green color and enabling photosynthesis. Magnesium is also used in lightweight alloys for things like bike frames and laptop casings, and its powerful bright white flash has made it a favorite for historical camera flashes and dazzling fireworks. Other members include Strontium (Sr), responsible for the vibrant red hues in fireworks and sometimes found in glow-in-the-dark paints, and Barium (Ba), which gives fireworks a striking green and is famously used in medical "barium meals" to help doctors see your digestive system on X-rays.
Must Read
Ready to start exploring? It's easier than you think! A great first step is simply to look around you. Can you spot objects that might contain calcium or magnesium? Think about your diet – what foods are rich in these minerals? When you next see a fireworks display, try to identify the colors and recall which alkaline earth metal might be responsible. You could even read the labels on your supplements or garden fertilizers to see if they list magnesium or calcium. Simply observing and asking "what's that made of?" is a fantastic start to understanding the chemistry that underpins our world.
By taking a moment to appreciate these common yet extraordinary elements, you’ll find that chemistry isn’t just something confined to laboratories. It’s a dynamic, exciting force shaping every aspect of our lives. So go ahead, enjoy uncovering the hidden superpowers of alkaline earth metals!




