What Is Alkali And Alkaline Earth Metals

Ever wondered what makes fireworks sparkle or why your antacids work? A big clue lies in two groups of elements: the alkali metals and the alkaline earth metals. They're like the rockstars of the periodic table, full of energy and ready to react!
Alkali Metals: The Wild Ones
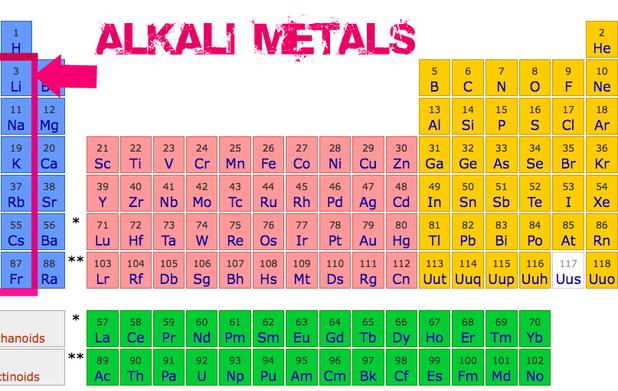
Think of the alkali metals as the over-enthusiastic puppies of the element world. They are found in the first group on the periodic table. Lithium, sodium, and potassium are all part of this lively bunch!
These guys are so eager to react that you won't find them hanging out in their pure form in nature. They're always attached to something else, like oxygen or chlorine, forming compounds. They're just too social to be alone!
Must Read
Explosive Personalities
Here's where it gets fun (and slightly dangerous, so don't try this at home!). When alkali metals meet water, things get… exciting. Really exciting. We are talking about explosions.
The reaction releases hydrogen gas, which then ignites, creating a burst of flame and a pop. The bigger the alkali metal, the bigger the bang. Imagine sodium throwing a party in a bathtub!
This reactivity is why alkali metals are stored in oil. It's like putting them in a time-out, preventing them from causing too much trouble. They are simply too reactive and needs to be tamed.
Everyday Superstars
Despite their explosive tendencies, alkali metals are incredibly useful. Sodium, for example, is a key component of table salt. Without it, our food would be pretty bland!
Lithium is a superhero in its own right. It's used in batteries that power our smartphones, laptops, and electric cars. Thank you, lithium, for keeping us connected!
Potassium is vital for plant growth and nerve function. It helps keep our hearts beating and our muscles working properly. We are deeply indebted to it.
Alkaline Earth Metals: The Slightly Calmer Cousins
Now, let's meet the alkaline earth metals. These elements reside in the second group on the periodic table. They're the alkali metals' slightly more mature and responsible cousins.
Magnesium, calcium, and barium are all members of this family. While still reactive, they're not quite as wild as their alkali metal relatives. They’re still eager to bond, but with a bit more finesse.
Strong and Sturdy
Alkaline earth metals are known for their strength and ability to form strong compounds. Calcium, for example, is essential for building strong bones and teeth.
Think of calcium as the construction worker of your body, constantly reinforcing your skeletal structure. Without it, we'd be a wobbly mess!
Magnesium is another workhorse, playing a crucial role in muscle and nerve function. It also helps regulate blood sugar levels. This is quite impressive!
Fireworks and Antacids: A Touch of Magic
Barium compounds are used to create vibrant green colors in fireworks. They are also used in medical imaging to help doctors see inside the body.
Magnesium hydroxide is a key ingredient in antacids. It neutralizes stomach acid, providing relief from heartburn. So, next time your stomach is acting up, thank the alkaline earth metals!
Calcium carbonate is another common antacid. It also helps build strong bones and teeth. These metals are multi-taskers.
Alkali vs. Alkaline Earth: What's the Difference?
So, what really sets these two groups apart? It boils down to their reactivity and the way they interact with other elements.
Alkali metals are more reactive because they have only one electron in their outermost shell. They're desperate to get rid of it to achieve a stable electron configuration.
Alkaline earth metals have two electrons in their outermost shell. They're still reactive, but not quite as desperate as the alkali metals.
Another key difference is their hardness. Alkali metals are soft and can be cut with a knife, while alkaline earth metals are harder and stronger.
Beyond the Periodic Table: A New Perspective
Hopefully, you now see alkali metals and alkaline earth metals in a new light. They're not just abstract concepts in a chemistry textbook.
They're essential components of our everyday lives, from the batteries that power our devices to the fireworks that light up our skies. They make up the table salt that flavors our food.
They are found in the medicine that eases our stomach aches. These elements are truly remarkable. Their properties benefit us in many different ways.
Next time you see a firework display or take an antacid, remember the alkali and alkaline earth metals. They're the unsung heroes of the chemical world, quietly working to make our lives better.
They are often misunderstood. We frequently take them for granted. Let us show appreciation for these reactive and essential elements.
Consider the fireworks that burst with vibrant green due to barium compounds, or the relief you feel after taking magnesium hydroxide for heartburn. These metals are incredibly helpful!
It's a fascinating story of reactivity, stability, and the surprising ways elements interact to shape our world. The periodic table is filled with wonder and potential.
So, embrace your inner chemistry enthusiast and continue to explore the elements that make up everything around us! The more you learn, the more you will love.
Who knew that something as seemingly dry as the periodic table could be so full of surprises and practical applications? Chemistry rocks!
