What Is A Semimetal On The Periodic Table
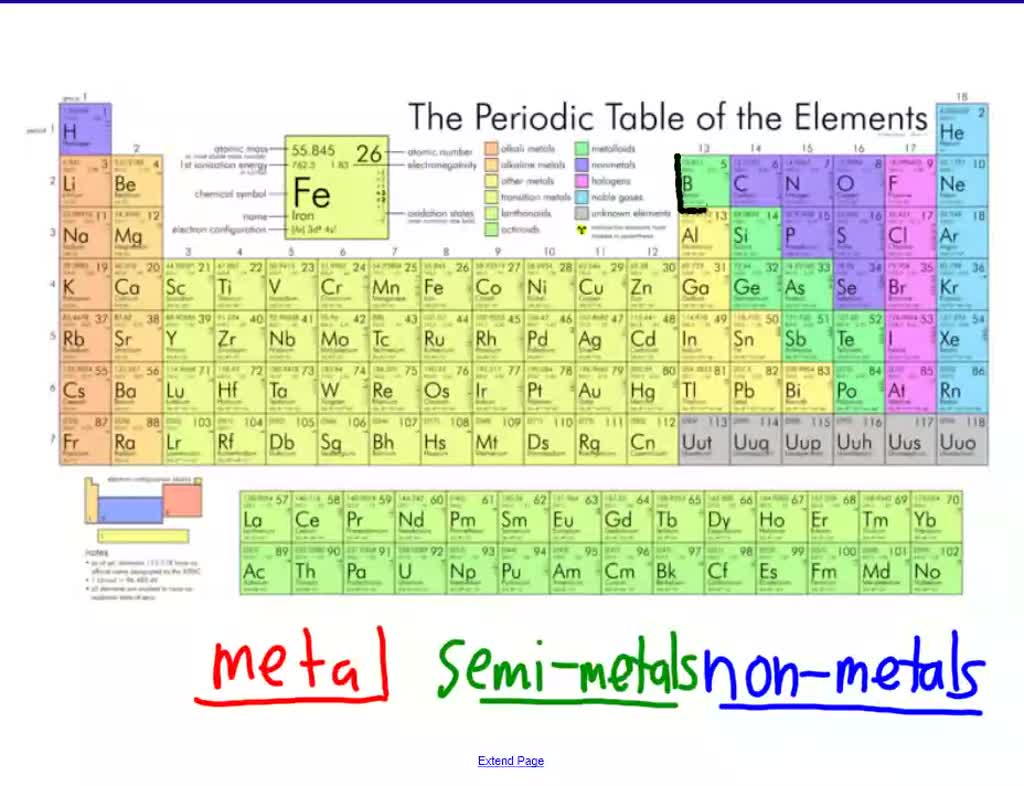
Hey friend! Ever looked at the periodic table and thought, "Wow, that's... a lot?" Yeah, me too. But today, let's zoom in on something super cool: semimetals! Also known as metalloids. Sounds kinda like a rejected Transformer, right?
What’s the Deal with These Semi-Guys?
Okay, so semimetals aren't quite metals. And they're definitely not nonmetals. They're like the indecisive friend who can't pick a restaurant. They have properties of both! Seriously, they're the chameleons of the element world.
Think of it like this: metals are the cool kids, conductors of electricity and heat. Nonmetals? Not so much. Semimetals are hanging out on the bleachers, sometimes joining in the metal party, sometimes chilling with the nonmetals. It all depends on the mood! Or, you know, the temperature and other stuff.
Must Read
There are usually six elements recognized as semimetals: boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), and tellurium (Te). Some scientists argue for others to be included, but these are the OG semi-crowd.
Why Should I Even Care?
Good question! Here's why: these guys are everywhere in technology. Seriously. Your phone? Semimetals. Your computer? Semimetals. The fancy solar panels on your neighbor's roof? Yep, probably semimetals.

Specifically, silicon is the rockstar of the semimetal world. It's the foundation of most computer chips. Without silicon, your TikTok scrolling would be a whole lot less smooth. Thank you, silicon!
Germanium, too, is used in transistors and semiconductors. Before silicon took over, germanium was the king of the tech world! Imagine the rivalry.
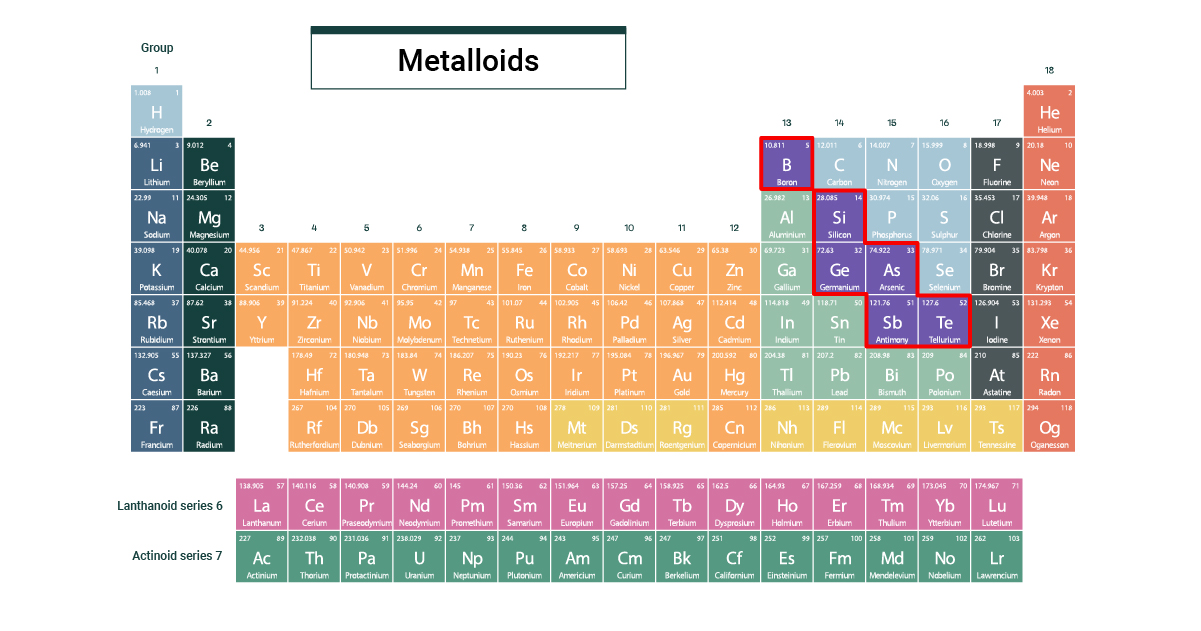
Quirky Facts and Fun Details
Ready for some mind-blowing stuff? Buckle up!
- Arsenic is poisonous! Don't go licking any arsenic rocks. It’s used in some pesticides and wood preservatives, but definitely keep it away from your snacks.
- Boron compounds are used in laundry detergent! Talk about a multi-tasker. From cleaning clothes to strengthening glass (think Pyrex!), boron is a busy bee.
- Tellurium is named after the Earth (Tellus in Latin). How cool is that? And get this: people who are exposed to tellurium compounds can develop "tellurium breath," which smells like garlic! Who needs vampires when you’ve got tellurium breath?
- Antimony was used in ancient Egypt as eye makeup! Talk about a bold look. It's also used as a flame retardant. So, thanks to antimony, your curtains might be a little less likely to burst into flames.
See? Semimetals aren't just some boring element group. They’re full of surprises!
Their Party Trick: Semiconductors
Okay, this is the coolest part. Semimetals are semiconductors. This means they can control how electricity flows through them. They can switch from being insulators (blocking electricity) to conductors (allowing electricity to flow) based on…stuff! Temperature, light, impurities… It’s like having a dimmer switch for electricity at an atomic level.

This "on-off" ability is what makes computers possible. Those tiny transistors in your phone? They use semiconductors to switch electrical signals on and off, representing the 0s and 1s of binary code. It's like a Morse code for machines, and semimetals are the operators!
Why Are They Called Semimetals and Not Metalloids?
Good question! Both terms are used, but "metalloid" is perhaps slightly more common and considered more descriptive. "Semimetal" highlights their conductivity properties, while "metalloid" emphasizes their intermediate nature between metals and nonmetals.

So, Are Semimetals Metals or Not?
That's the million-dollar question, isn't it? The answer is... it depends! It depends on the specific semimetal, the temperature, the pressure, and a whole bunch of other factors. They're complicated! But that's what makes them so interesting. They defy easy categorization.
They're the rebels of the periodic table, the cool kids who don't follow the rules. And without them, our modern world wouldn't be the same. So next time you're using your phone or computer, take a moment to appreciate the amazing, versatile, and slightly confusing world of semimetals!
Keep exploring! The periodic table is full of surprises. Who knows what other elemental wonders you'll discover?
