What Is A Glass Transition Temperature

Alright, gather 'round, friends! Let's talk about something mind-boggling, yet surprisingly relevant to your everyday life: The Glass Transition Temperature (Tg). Now, I know what you're thinking: "Sounds like something I slept through in high school chemistry." But trust me, this is way more exciting than balancing equations – and way less likely to give you a headache. Unless, you know, you really think about it hard. Then maybe a little headache.
Imagine you're making caramel. You start with sugar, a nice, crystalline, well-behaved solid. Heat it up, and BAM! It melts into a syrupy, gooey liquid. Easy peasy. That's a pretty straightforward phase transition: solid to liquid at a clear melting point. But what if, instead of caramelizing, you were trying to make...super-strong, un-shatterable candy? That’s where Tg comes in.
So, What's the Big Deal?
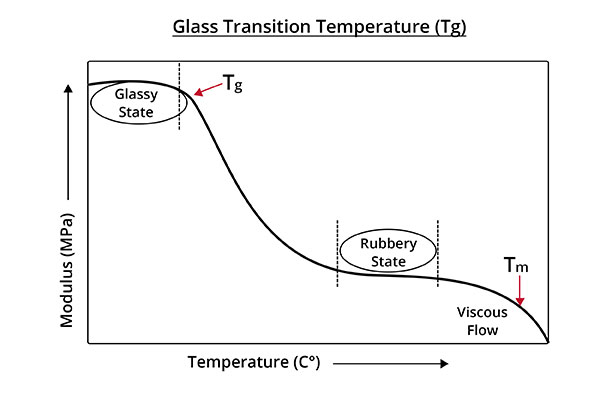
The Tg is basically the temperature at which a material goes from being a relatively rigid, glassy solid to a more rubbery or viscous state. It's like the material can't decide if it wants to be a solid or a liquid, so it just hangs out in this awkward in-between zone. Think of it as a material’s mid-life crisis. It's not quite solid, not quite liquid...it's complicated!
Must Read
Now, here's the kicker: It's not a "melting point". Oh no, that would be too easy! Melting points are for crystalline solids, things with nice, orderly arrangements of molecules. Glassy materials, on the other hand, are amorphous. Think of them as the rebels of the molecular world. No rules, no order, just a bunch of molecules doing their own thing. They’re the hipsters of the material science world, refusing to conform to boring crystalline structures.
Because there's no neat and tidy crystal structure to break down, glassy materials don't melt at a specific temperature. Instead, they soften gradually as they approach their Tg. It's like watching a grumpy old man slowly start to loosen up after a few cups of coffee (or maybe something stronger...we don't judge).

Everyday Examples That Will Blow Your Mind (Maybe)
You're probably surrounded by materials that have a Tg right now. Plastic is a huge one! That Tupperware container you use to store leftovers? It has a Tg. And if you put it in the freezer and then try to bend it, you might find it cracks. Why? Because the freezer dropped the plastic's temperature below its Tg, making it brittle and prone to shattering. Oops! Hope that wasn’t your grandma's favorite container.
Polymers are particularly fond of having a Tg. Polymers are long chains of molecules, like spaghetti noodles all tangled together. The Tg affects how flexible or rigid those "noodles" are. This is why some plastics are bendy and some are stiff. It all comes down to their Tg and the temperature you're using them at.

Think about rubber. It's super stretchy and flexible at room temperature. That’s because its Tg is usually way below room temperature. But if you cool it down enough (say, by sticking it in liquid nitrogen – please don’t actually do this without proper safety gear!), it will become hard and brittle. You could probably shatter it with a hammer! (Again, don't actually do this...unless you're a scientist with a grant and a penchant for destruction.)
Why Should You Care? (Besides the Impending Tupperware Disaster)
Understanding the Tg is crucial in all sorts of industries. Material scientists use it to design everything from airplane parts to medical implants. Want to make a plastic that can withstand the extreme temperatures of space? You need to choose a material with a high Tg. Need a rubber that stays flexible in freezing conditions? You need a material with a low Tg. It's all about choosing the right material for the job.
And hey, next time you're struggling to open a stubborn jar lid, remember the Tg. Running the lid under hot water might just raise the temperature of the plastic gasket above its Tg, making it more flexible and easier to grip. See? Science to the rescue! (Although, if that doesn’t work, you might just need a bigger friend.)
So, there you have it! The Glass Transition Temperature, demystified. It's not just some obscure scientific concept; it's a fundamental property of materials that affects our everyday lives in countless ways. Now go forth and impress your friends with your newfound knowledge! And maybe buy some better Tupperware.
Bonus Fact: Scientists are still debating the exact nature of the glass transition. It's one of those problems that's simple to understand but incredibly difficult to explain fully. So, if you're looking for a career path that's both challenging and endlessly fascinating, consider becoming a material scientist! Just be prepared for a lot of head-scratching and the occasional Tupperware-related crisis.

