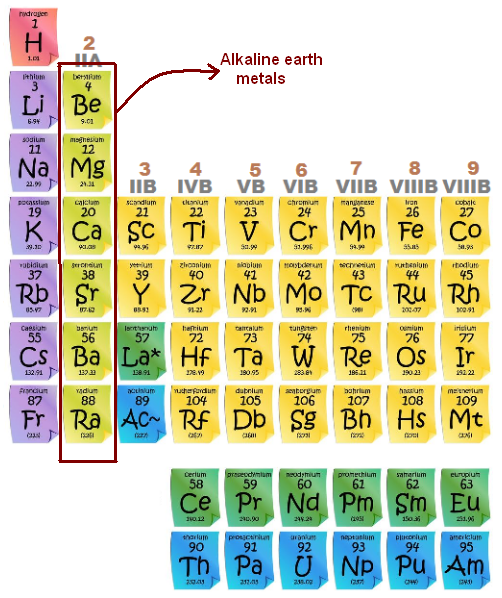
What Group Is Alkaline Earth Metals
Ever wonder what makes fireworks glow with such vibrant colors, or how your antacids soothe a fiery stomach? A big part of the answer lies in a fascinating family of elements known as the alkaline earth metals. Learning about them isn't just about chemistry class; it's about understanding the world around us in a richer, more exciting way. Think of it as unlocking a secret code to the building blocks of matter!
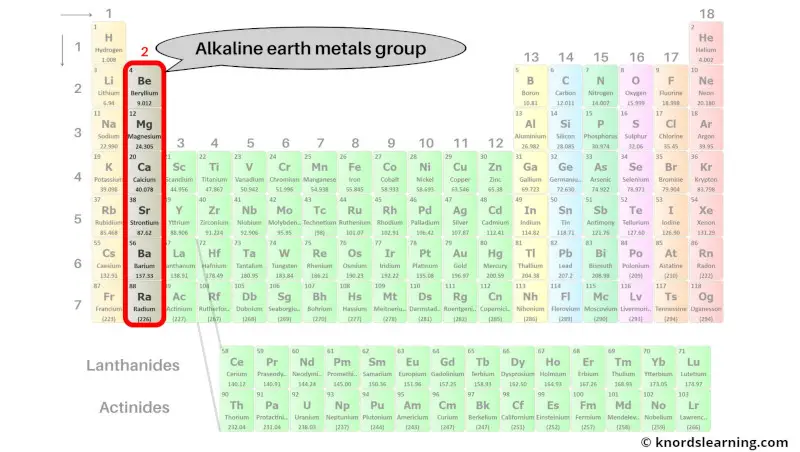
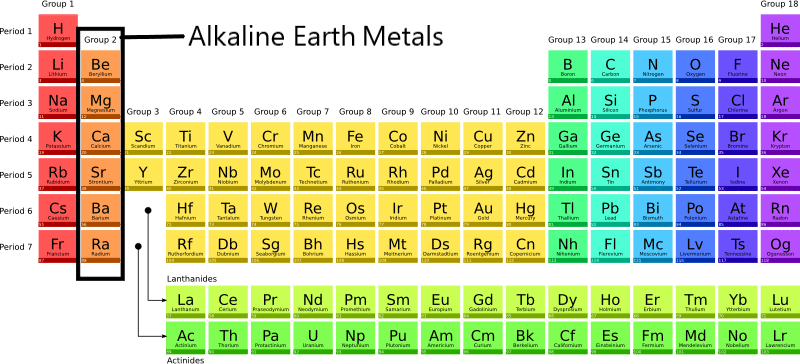
So, what exactly are these alkaline earth metals? They're a group of six elements nestled in the second column (Group 2) of the periodic table: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). Each of these elements shares similar chemical properties, making them a tight-knit family. They're all silvery-white, relatively soft (though harder than the alkali metals), and quite reactive – though less so than their Group 1 neighbors.
The "alkaline earth" name comes from the fact that their oxides (compounds with oxygen) form alkaline, or basic, solutions when dissolved in water. "Earth" refers to the fact that these oxides were historically difficult to melt and were often found in the earth’s crust. But their purpose extends far beyond just making alkaline solutions. Their real value lies in their diverse applications, thanks to their unique chemical behavior.
Must Read
What are the benefits of understanding these metals? Well, for starters, it helps you appreciate the roles they play in our daily lives. Calcium, for example, is essential for strong bones and teeth. It's also crucial for muscle function and nerve transmission. Without enough calcium, our bodies simply wouldn't work properly. Magnesium is another vital nutrient involved in hundreds of biochemical reactions in our bodies, from energy production to muscle relaxation. Deficiencies in magnesium are linked to various health problems.
Beyond human health, these elements are used in countless other applications. Magnesium is a key component in lightweight alloys used in aerospace and automotive industries. Strontium is responsible for the brilliant red color in fireworks. Barium compounds are used in medical imaging, such as barium swallows, to help visualize the digestive tract. Even radium, though radioactive and less commonly used now, was once employed in cancer treatment.
Learning about alkaline earth metals doesn't have to be confined to textbooks. Here are a few simple ways to explore them: Look at the nutrition labels of your food to see how much calcium and magnesium they contain. Research different types of fireworks and the elements responsible for their colors. If you're feeling adventurous, you could even try growing plants in different types of soil to see how calcium levels affect their growth. Explore the geology of your local area, looking for rocks and minerals containing alkaline earth metals. There are also many safe and engaging online resources, like interactive periodic tables and educational videos, that can bring these elements to life.
The alkaline earth metals are more than just elements on a chart; they're integral parts of the world around us. By understanding their properties and applications, we gain a deeper appreciation for the chemistry that shapes our lives. So, next time you eat a calcium-rich meal or marvel at a colorful firework display, remember the amazing alkaline earth metals and the fascinating science behind them.