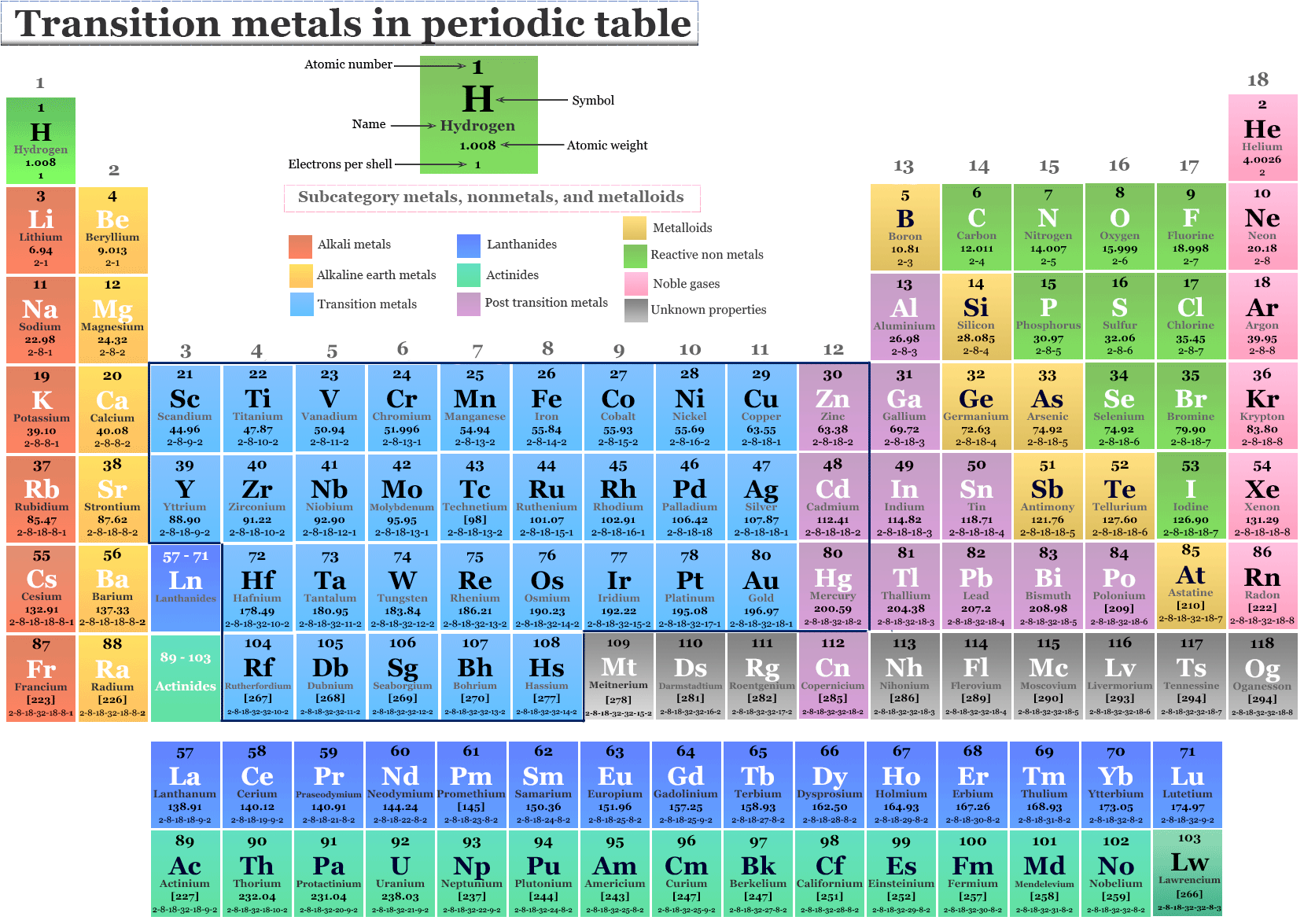
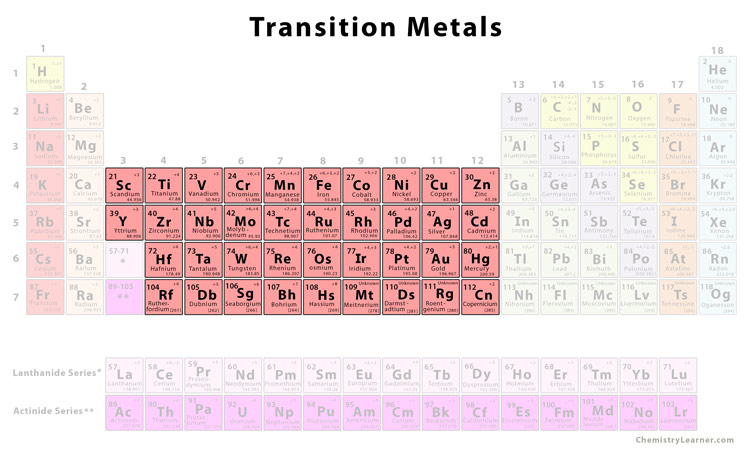
What Elements Are In The Transition Metals
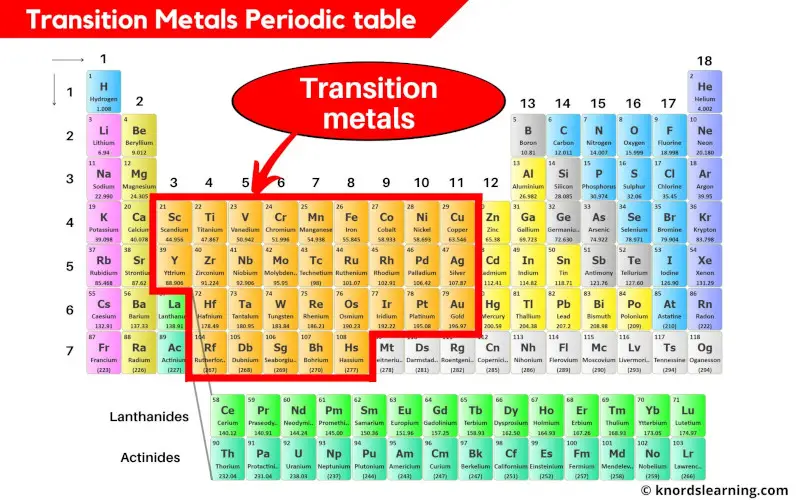
Ever wonder what makes your phone screen glow, or what gives jewelry its sparkle? The answer, in part, lies with a fascinating group of elements called the transition metals. They're not just tucked away in labs; they're essential to our modern world, and understanding them opens up a whole new appreciation for the chemistry around us.
So, what are the transition metals? Simply put, they're a block of elements found in the middle of the periodic table, specifically in groups 3 through 12. But it's not just their location that makes them special; it's their unique electronic structure. Unlike elements in other groups, transition metals have partially filled d orbitals, which give them a remarkable ability to form compounds with multiple oxidation states (fancy chemistry speak for having different charges) and create vibrant colors.
The beauty of transition metals lies in their versatility. Take iron (Fe), for example. It's the backbone of our infrastructure, forming the steel used in bridges, buildings, and cars. Copper (Cu), another essential transition metal, is a superb conductor of electricity, making it indispensable for wiring and electronics. And what about titanium (Ti)? Its strength and lightweight properties make it ideal for aircraft and medical implants.
Must Read
But the impact of transition metals extends far beyond industry. In education, they serve as excellent examples of chemical principles. Observing the different colors of transition metal compounds in solution is a classic chemistry lab activity, illustrating the relationship between electronic structure and light absorption. Think of copper sulfate solution, which has that iconic bright blue color. That vibrant hue is directly linked to copper's unique electron configuration.
Even in daily life, you encounter transition metals in surprising places. The pigments that give paint its color often contain transition metal oxides. For instance, chromium (Cr) is used to create green pigments, and cobalt (Co) contributes to blue hues. Catalytic converters in cars, which reduce harmful emissions, rely on transition metals like platinum (Pt), palladium (Pd), and rhodium (Rh) to speed up chemical reactions.
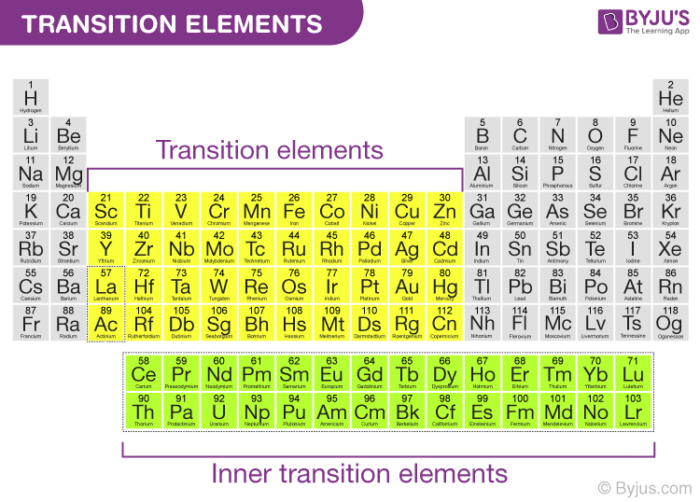
So, how can you explore the world of transition metals further? One simple way is to pay closer attention to the labels of products you use every day. Look for ingredients like "iron oxide" in cosmetics or "titanium dioxide" in sunscreen. These are clues to the presence of transition metals and their unique properties. Another fun activity is to collect different types of rocks and minerals. Many minerals owe their colors to trace amounts of transition metals.
You can also find countless resources online, including interactive periodic tables and educational videos that delve deeper into the chemistry of transition metals. The key is to cultivate a sense of curiosity and to remember that chemistry isn't confined to textbooks; it's all around us, shaping the world in ways both big and small. So, the next time you admire a piece of jewelry or drive across a bridge, take a moment to appreciate the remarkable elements that make it all possible – the versatile and essential transition metals.