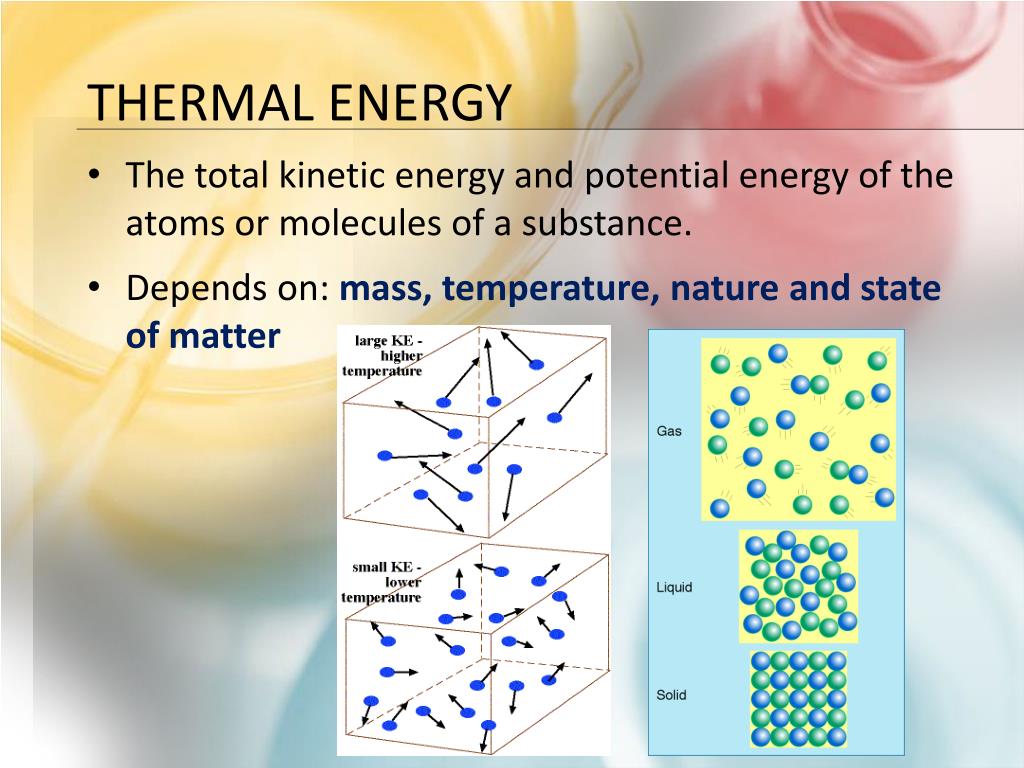
What Does Thermal Energy Depend On
Have you ever wondered why your grandma's freshly baked cookies burn your tongue way more than a sip of lukewarm tea, even if you took the tea first? Or why a tiny sparkler can seem harmless while a roaring bonfire radiates enough heat to melt your eyebrows? The answer, in a nutshell, lies in understanding what thermal energy depends on.
Think of thermal energy as the chaotic dance party happening inside everything around you. All those tiny particles, atoms and molecules, are constantly jiggling, wiggling, and bouncing off each other. The faster they move, the more energetic (and hotter!) things get. But it's not just about speed; it's also about how many dancers are on the floor.
Size Matters (Sometimes)
Let's say you have two cups of water. One is a teacup, and the other is a giant mug you found at a garage sale that probably belonged to a coffee-addicted giant. Both cups are filled with water heated to the same temperature. Which one has more thermal energy? The giant mug, hands down! Even though the water in both cups is at the same temperature, the mug holds way more water, meaning there are way more water molecules bouncing around. More dancers, more energy! It's like a stadium concert versus a small club gig. Same music, vastly different energy.
Must Read
Now, this is where it gets a little tricky. Imagine a tiny sparkler versus that bonfire. The sparkler might have a super high temperature, like really really high, but it doesn't have a lot of "stuff" (mass) to it. The bonfire, on the other hand, is massive. It’s a mountain of fuel that is burning. So while the sparkler might sting if you touch it, the bonfire can actually cause some serious damage. Why? Because of the total amount of thermal energy stored.

The Temperature Tango
Okay, so we know quantity matters, but what about speed? That's where temperature comes in. Temperature is basically a measure of the average speed of those dancing particles. Higher temperature? Faster dancing! If you have a tiny amount of something at a super high temperature, like a welding arc, it can still deliver a concentrated burst of thermal energy, even though the actual amount of "stuff" involved is small. Think of it as a tiny but incredibly fast army of super-charged particles.
Imagine a single, super-fast ping pong ball versus a thousand slowly rolling bowling balls. The ping pong ball has a lot of kinetic energy because of its speed. But the bowling balls, collectively, have way more. That's kind of like temperature versus quantity in the world of thermal energy.

What is it Made Of?
Believe it or not, what something is made of also plays a role. Different materials require different amounts of energy to heat up. This is called specific heat capacity. Water, for example, has a high specific heat capacity. It takes a lot of energy to raise the temperature of water, which is why it's used as a coolant in car engines. Metal, on the other hand, heats up much faster. That's why a metal spoon left in a hot pot will get burning hot way quicker than the water itself.
"Thermal energy is the sum of all the kinetic energy of the particles in a system, so temperature and the amount of substance are factors, but specific heat matters too! Remember that energy can be transferred and transformed."
So, next time you're enjoying a cup of tea, remember the chaotic dance party happening at the molecular level. Think about the temperature, the quantity of the tea, and even the material of the cup. They all play a part in determining how much thermal energy is packed into that comforting, warming drink. And maybe, just maybe, you'll appreciate that cup of tea just a little bit more, knowing that it's not just a beverage, but a universe of tiny, energetic dancers working to keep you warm.


