What Does The Number Next To Isotopes Signify
Okay, so you've probably heard about isotopes, maybe in a science class that felt like it lasted for seventeen years, or perhaps during a particularly intense episode of your favorite science show. But what's with that number hanging around next to them? Like Carbon-14. What does the 14 mean?! Don't worry, we're about to break it down, no PhD required. Think of it as the secret handshake of the atomic world.
Imagine this: you're at a family reunion. Everyone's a "Smith," but there's "Uncle Joe Smith," "Cousin Sarah Smith," and "Great-Grandma Mildred Smith." They all share the same last name (Smith, like an element!), but they're all a little different. Isotopes are similar. They're all the same element, but they have slightly different "weights." That number tells you about that "weight."
The Secret's in the Nucleus
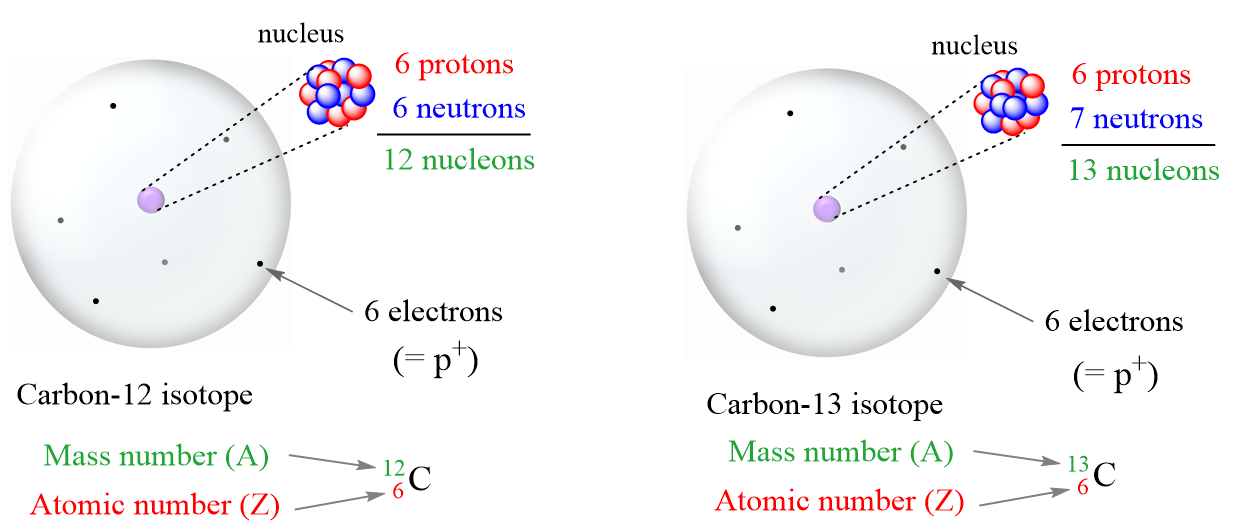
Deep inside every atom is the nucleus, the atom's tiny but powerful core. This is where the party's really happening. The nucleus contains two types of particles: protons and neutrons. Protons are like the VIPs; they determine what element it is. If it has one proton, it's hydrogen. Six protons? It's carbon. Eight? Oxygen. You get the idea. Change the number of protons, and you change the whole element!
Must Read
Neutrons, on the other hand, are the... let's call them "chill guests." They don't change the element's identity, but they do affect its mass. Think of it this way: protons are like the band at a party. Without them, there's no music, no party. Neutrons are the folks who bring extra snacks. They make the party a little bigger, but it's still the same party.
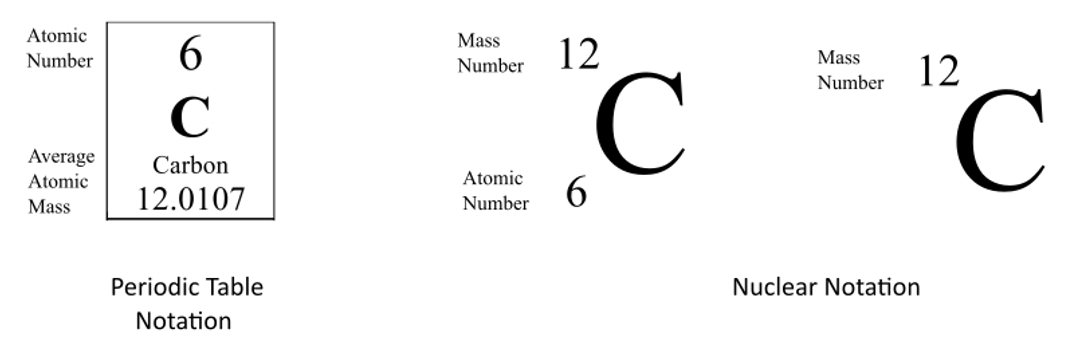
The number next to the isotope name (like that "14" in Carbon-14) is the mass number. It's simply the total number of protons and neutrons in the nucleus. So, if Carbon-14 has a mass number of 14, that means it has 6 protons (because all carbon atoms have 6 protons) and 8 neutrons (14 - 6 = 8).

Why Does It Matter?
Okay, so we know what the number means, but why should you care? Well, these differences in neutron count can drastically change an isotope's behavior. Some isotopes are stable, like Great-Grandma Mildred, calmly knitting in the corner. They're perfectly content to stay as they are. Others are unstable, like Uncle Joe after a few too many glasses of punch. They're radioactive and spontaneously break down, emitting energy and particles.
Think of it like a tower of blocks. If you've stacked the blocks just right, the tower is stable and stands tall. But if you add too many blocks unevenly (extra neutrons!), the tower becomes unstable and eventually topples over (radioactive decay!).

This radioactivity can be both a blessing and a curse. Carbon-14, for example, is radioactive and used for carbon dating. This allows scientists to figure out how old fossils and ancient artifacts are, essentially telling us when those ancient civilizations had their family reunions! On the flip side, radioactive isotopes can be dangerous if not handled properly, like a rogue sparkler at a kid's birthday party.
Examples in Everyday Life
You might not realize it, but isotopes are everywhere! Iodine-131 is used in medical treatments to treat thyroid problems. Uranium-235 is used in nuclear power plants to generate electricity. Deuterium (Hydrogen-2), often called "heavy hydrogen," is used in nuclear research and sometimes in specialized chemical reactions.
So, the next time you hear about isotopes, don't run screaming for the hills. Remember the family reunion analogy! The number next to the isotope is just telling you how many "snacks" (neutrons) are at the party. It's the secret to understanding their behavior and how they impact the world around us. And who knows, maybe you’ll impress someone at your next cocktail party with your newfound knowledge of atomic nuclei. Just try not to get too radioactive!