What Are Transition Elements On The Periodic Table
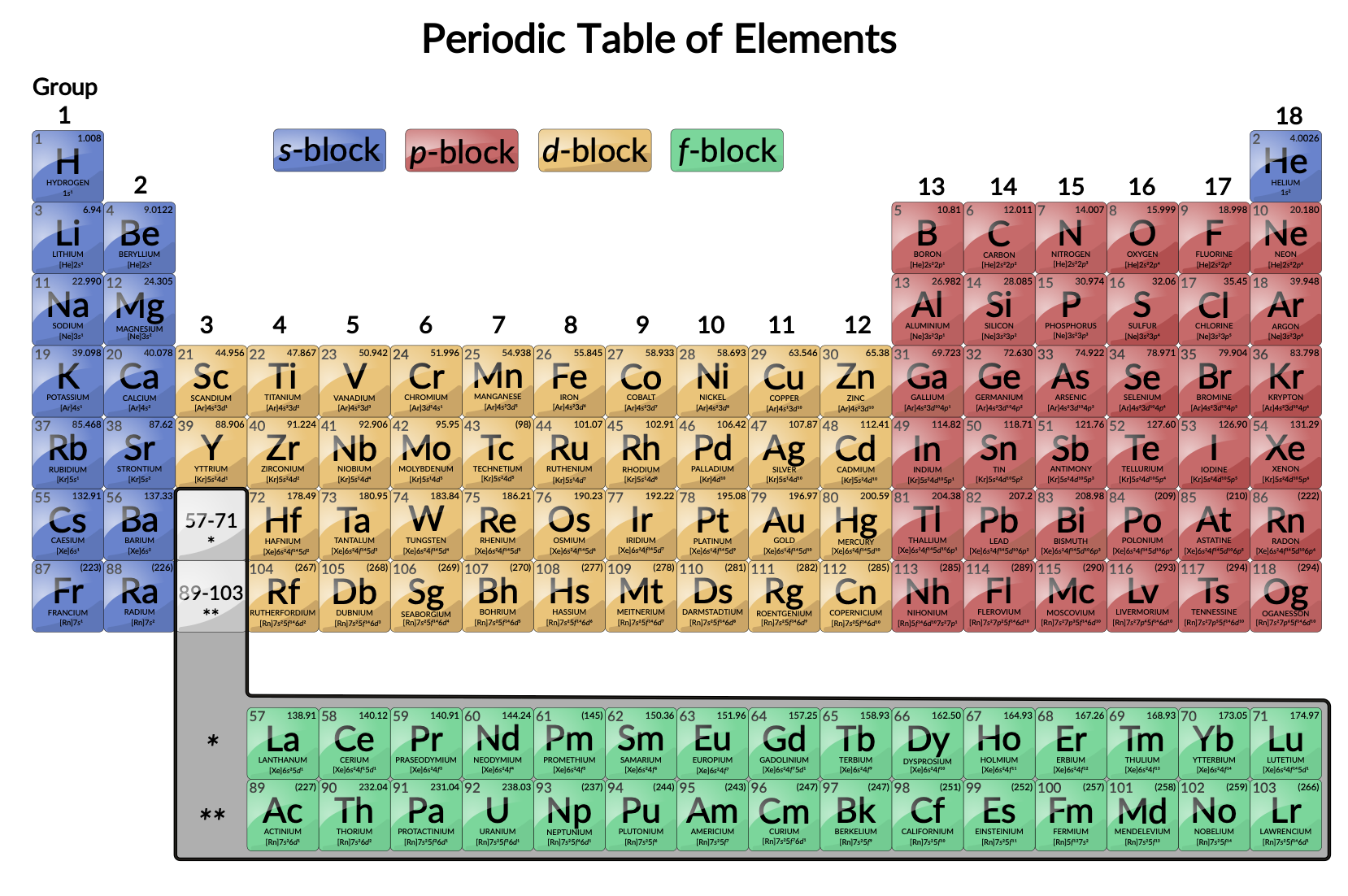
Ever glanced at the periodic table and felt a sudden urge to nap? We get it. It can look intimidating. But fear not, intrepid explorer of everyday science! We're diving into a fascinating section called the transition elements – and trust us, they're way cooler than they sound.
What's the Deal with Transition Elements?
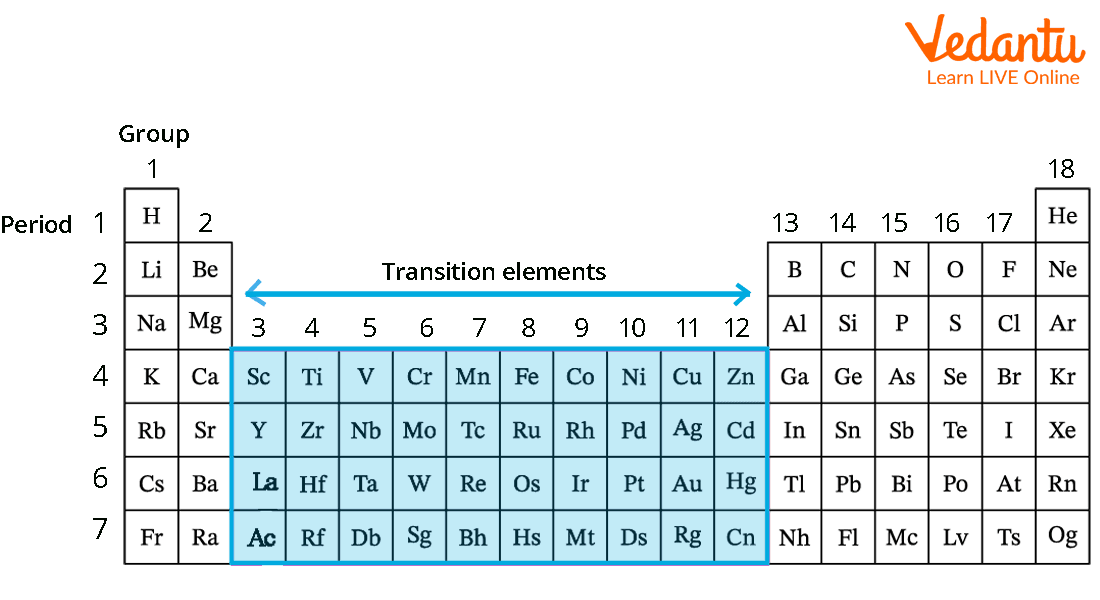
Think of the periodic table as a neatly organized apartment building. The transition elements are the residents living smack-dab in the middle, in what chemists affectionately call the "d-block." They're the rebels of the atomic world, not quite conforming to the rigid rules of their neighbors on either side. Their defining characteristic? They can form multiple positive ions, meaning they can happily bond with various other elements in multiple ways. Think of it as being super versatile in the dating world!
Quick Chemistry Refresher: Remember those electron shells buzzing around the nucleus of an atom? Transition elements are special because their outermost electron shells aren't fully filled. This is what gives them their funky properties and makes them so useful.
Must Read
The Rockstar Elements of the Group
Let's meet some of the stars! We're talking about elements like:
- Iron (Fe): The backbone of steel, construction, and even your blood (hemoglobin, to be precise). Think of it as the dependable best friend who always has your back.
- Copper (Cu): Essential for electrical wiring and plumbing, it's the silent workhorse of modern infrastructure. Also, it’s what makes the Statue of Liberty green!
- Gold (Au): The ultimate symbol of wealth and status. From ancient artifacts to modern jewelry, gold has captivated humanity for millennia. It's the Beyoncé of elements – eternally fabulous.
- Silver (Ag): Another precious metal, prized for its conductivity and antimicrobial properties. It’s used in everything from electronics to photography. Think of it as the sophisticated artist of the elements.
- Titanium (Ti): Lightweight yet incredibly strong, titanium is the superhero of materials. Used in airplanes, medical implants, and high-performance sports equipment, it's the element that lets us soar.
These are just a few examples. The list goes on! Each transition element has its unique set of superpowers.
Why Should You Care? Everyday Applications
Okay, so they're chemically interesting. But how do transition elements impact your daily life? Let us count the ways:
- Catalysis: Many transition metals act as catalysts, speeding up chemical reactions without being consumed themselves. This is crucial in manufacturing everything from plastics to pharmaceuticals. Think of them as the matchmakers of the chemical world.
- Pigments: The vibrant colors you see around you often come from transition metal compounds. From the red in your lipstick (iron oxide) to the blue in your favorite painting (cobalt), these elements bring color to our lives.
- Electronics: Copper wiring in your home, gold connectors in your computer, and silver in your smartphone – transition elements are essential for modern technology. They're the unsung heroes of the digital age.
- Medicine: Platinum-based drugs are used to treat cancer, and iron supplements help combat anemia. Transition elements are literally lifesavers.
Even that stainless steel cookware you use every day owes its corrosion resistance to the presence of chromium, a transition metal!
A Little Cultural Chemistry
Did you know that the alchemists of old were obsessed with trying to turn base metals like lead into gold? While they never succeeded (sorry, folks, alchemy isn't real), their experiments laid the foundation for modern chemistry and our understanding of transition elements. Talk about dedication to a dream!
The term "noble metal" (referring to gold, platinum, and other elements that resist corrosion) reflects their historical importance and perceived value. These elements have been prized for their beauty and durability for centuries, appearing in countless works of art, jewelry, and currency.

Practical Tip: Identifying Transition Metals
One easy way to spot a transition metal compound is by its color. Many of them produce brightly colored solutions and solids. Think of the vibrant greens of copper salts or the deep blues of cobalt compounds. This isn't a foolproof method, but it's a fun trick to impress your friends (or at least win at trivia night!).
Bringing it Home: A Touch of Chemistry in Your Day
Next time you admire a piece of jewelry, use your phone, or simply cook a meal, take a moment to appreciate the crucial role that transition elements play in our modern world. They're the hidden ingredients that make so much of our lives possible, and appreciating them adds a new layer of wonder to the everyday. The periodic table isn’t just a chart – it’s a map to understanding the very fabric of reality, and the transition elements are a vital and vibrant part of that story.

