What Are The Physical Properties Of A Metal

Okay, let's talk metal. Not headbanging metal, though that's cool too. We're diving into the stuff metal is made of. The things that make it, well, metal.
Shine Bright Like a... Metallic Surface
First up: luster. That's just a fancy word for shine, right? Metals are famous for their glow. Think of your grandma's silverware. Or, you know, a shiny new penny.
Unpopular opinion: polised plastic is almost the same thing. I mean, almost. But the real deal is, metals have a special way of reflecting light. Makes them look all fancy and expensive.
Must Read
The Hammer Test: Malleability
Next, malleability. Try saying that three times fast! Basically, it means you can smack a metal into a thin sheet.
Think aluminum foil. Or a blacksmith hammering away at a sword. My theory? Blacksmiths are just metal therapists. Relieving its stress.
Gold is like, the king of malleable metals. You can make it crazy thin. Like, see-through thin! It's insane.
Stretching the Truth: Ductility
Ductility is like malleability's skinny cousin. It means you can stretch metal into a wire. Copper wires are everywhere. In your walls, in your phone, everywhere!
Imagine trying to make a plastic wire. Snap! Game over. Metal just keeps going and going. Like the Energizer bunny, but made of copper.
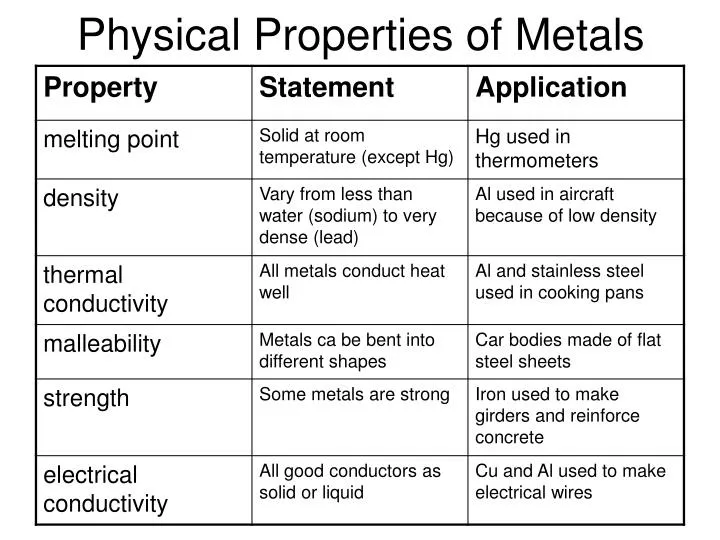
Feeling the Heat: Thermal Conductivity
Okay, let's talk heat. Metals are thermal conductors. That means they let heat pass through them easily. Think about a metal spoon in a hot cup of coffee.
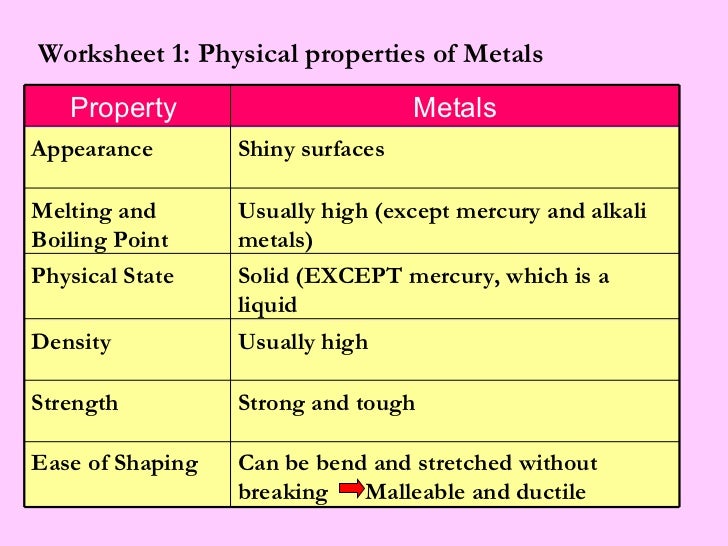
Ouch! Burns your hand, right? That's the metal doing its job. Transferring all that lovely heat. Maybe not so lovely for your fingertips, though.
Unpopular opinion: hot chocolate tastes better with a metal spoon anyway. Fight me.
Getting a Charge: Electrical Conductivity
Electricity! Zzzzap! Metals are also electrical conductors. That's why they're used in wiring. It's how your lights turn on, and your TV works.
Imagine trying to power your house with a rubber band. Chaos! Metals are the unsung heroes of modern life. We owe them a lot.
But why? The Electron Sea!
Here's a little science lesson. Inside a metal, the atoms share their electrons. It's like a big electron party. A free-for-all.
This "sea" of electrons is what allows the metal to conduct heat and electricity so well. They're just zipping around, passing the energy along.
The Strength Test: Tensile Strength
How strong is metal? Really strong! Tensile strength is its ability to resist being pulled apart. Think bridges, skyscrapers, and... chains!

Metals can hold a lot of weight. Which is good, because nobody wants their bridge collapsing. It's bad for traffic.
Density: How Heavy Is That Thing?
Density is how much stuff is packed into a certain space. Metals are generally pretty dense. Pick up a small metal ball. It's surprisingly heavy!
Lead is super dense. That's why it's used in weights and stuff. Gold is also really dense. Makes those gold bars seem extra valuable.
The Magnetic Personality: Ferromagnetism
Some metals are magnetic! Iron, nickel, and cobalt are the big players here. They're ferromagnetic. That means they can be magnetized.
Think magnets sticking to your fridge. Or the needle of a compass pointing north. Magic! (Okay, not really magic. But still cool.)
Unpopular opinion: fridge magnets are the best souvenirs. Fight me.

Melting Point: Things Get Hot
Melting point is the temperature at which a metal turns into a liquid. Some metals melt at low temperatures. Others need a lot of heat.
Mercury is liquid at room temperature. That's why it's used in old-fashioned thermometers. Tungsten has a super high melting point. That's why it's used in light bulb filaments.
Imagine trying to melt tungsten with a candle. Good luck with that!
Hardness: Tough Stuff
Hardness is a metal's ability to resist being scratched or dented. Diamonds are the hardest material on Earth. But some metals are pretty tough too.
Titanium is super strong and hard. That's why it's used in aerospace and medical implants. It's basically the Wolverine of metals.
Corrosion Resistance: Fighting the Rust
Corrosion is when a metal reacts with its environment and starts to break down. Rust is a form of corrosion that affects iron.
Some metals are more resistant to corrosion than others. Stainless steel is awesome because it doesn't rust easily. That's why it's used in kitchen sinks and cookware.
Unpopular opinion: a little rust gives things character. Adds that "antique" charm, you know?
Metal Alloys: Mixing It Up
What happens when you mix two or more metals together? You get an alloy! Alloys often have better properties than the individual metals.
Steel is an alloy of iron and carbon. It's stronger and more durable than pure iron. Brass is an alloy of copper and zinc. It's used in musical instruments and plumbing fixtures.
Alloys are like the Avengers of the metal world. Teamwork makes the dream work!
So, What Are The Physical Properties Of Metal?
So there you have it! Luster, malleability, ductility, conductivity, strength, density, magnetism, melting point, hardness, and corrosion resistance. That's metal in a nutshell.
Metals are fascinating materials with a wide range of properties. They're essential to our modern world. So next time you see a piece of metal, take a moment to appreciate its awesomeness.
Now, if you'll excuse me, I'm going to go polish my spoon. It's looking a little dull.
