What Are All The Transition Metals
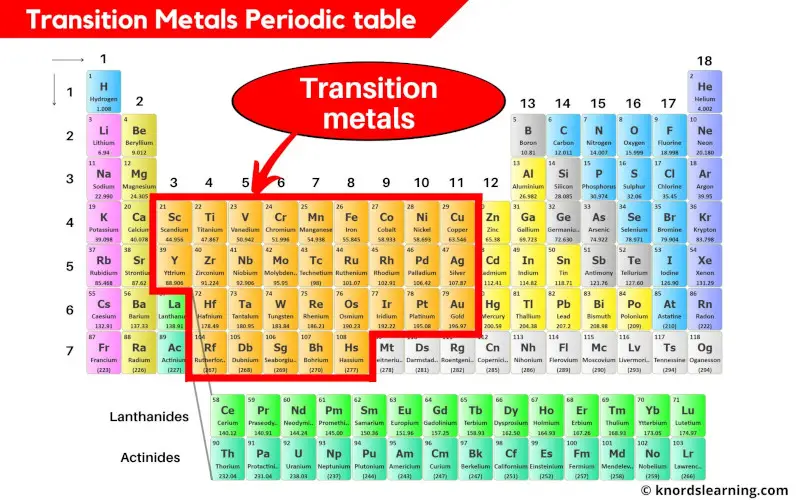
Ever heard of the Transition Metals? They're not some secret society of metals holding clandestine meetings, although that does sound intriguing.
Instead, they're a group of elements chilling out in the middle of the periodic table. Think of them as the cool kids of the element world, known for their vibrant colors and versatile personalities.
Meet the Metal Squad
Imagine the periodic table as a seating chart at a giant element party. On the left, you've got the alkali and alkaline earth metals, all eager to react. Over on the right, the halogens are busy being reactive and a bit clingy.
Must Read
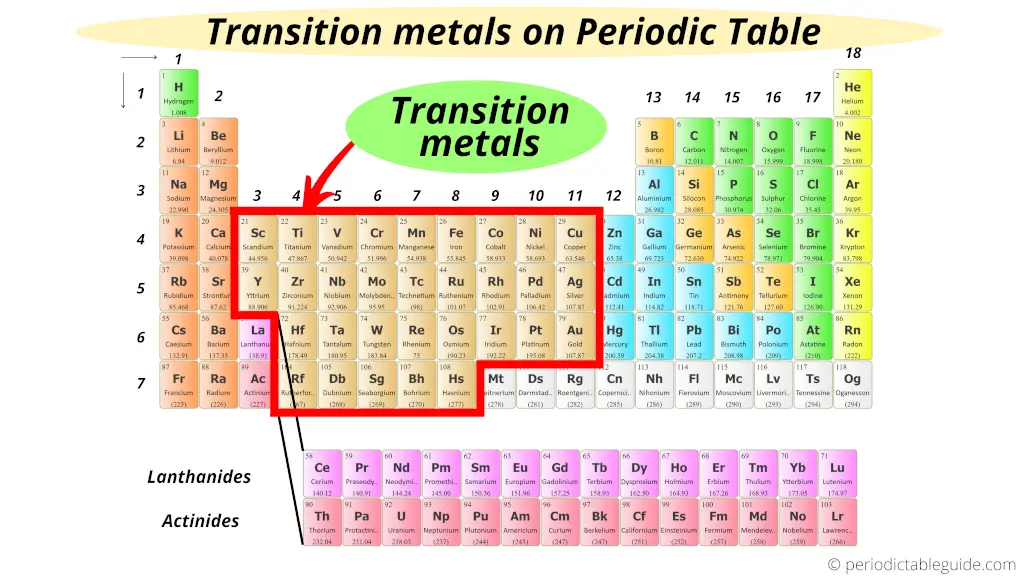
But right smack-dab in the middle? That's where the Transition Metals are holding court!
These guys include familiar names like iron (Fe), copper (Cu), gold (Au), and silver (Ag). They also feature less-household names like titanium (Ti), zinc (Zn), and chromium (Cr). It's a big, diverse crew!
What Makes Them So Special?
So, what sets these metals apart from the rest of the elemental crowd? Well, they're all about having partially filled d-orbitals. Don't worry too much about what that means!
Just know it’s the reason for their superpowers. Namely, their ability to form colorful compounds and act as amazing catalysts.
Think of it like this: their d-orbitals are like little pockets, ready to hold electrons. It's this flexibility that gives them their unique properties.
A Rainbow of Applications
The Transition Metals aren't just sitting around looking pretty (though they are good at that). They're workhorses in all sorts of industries.
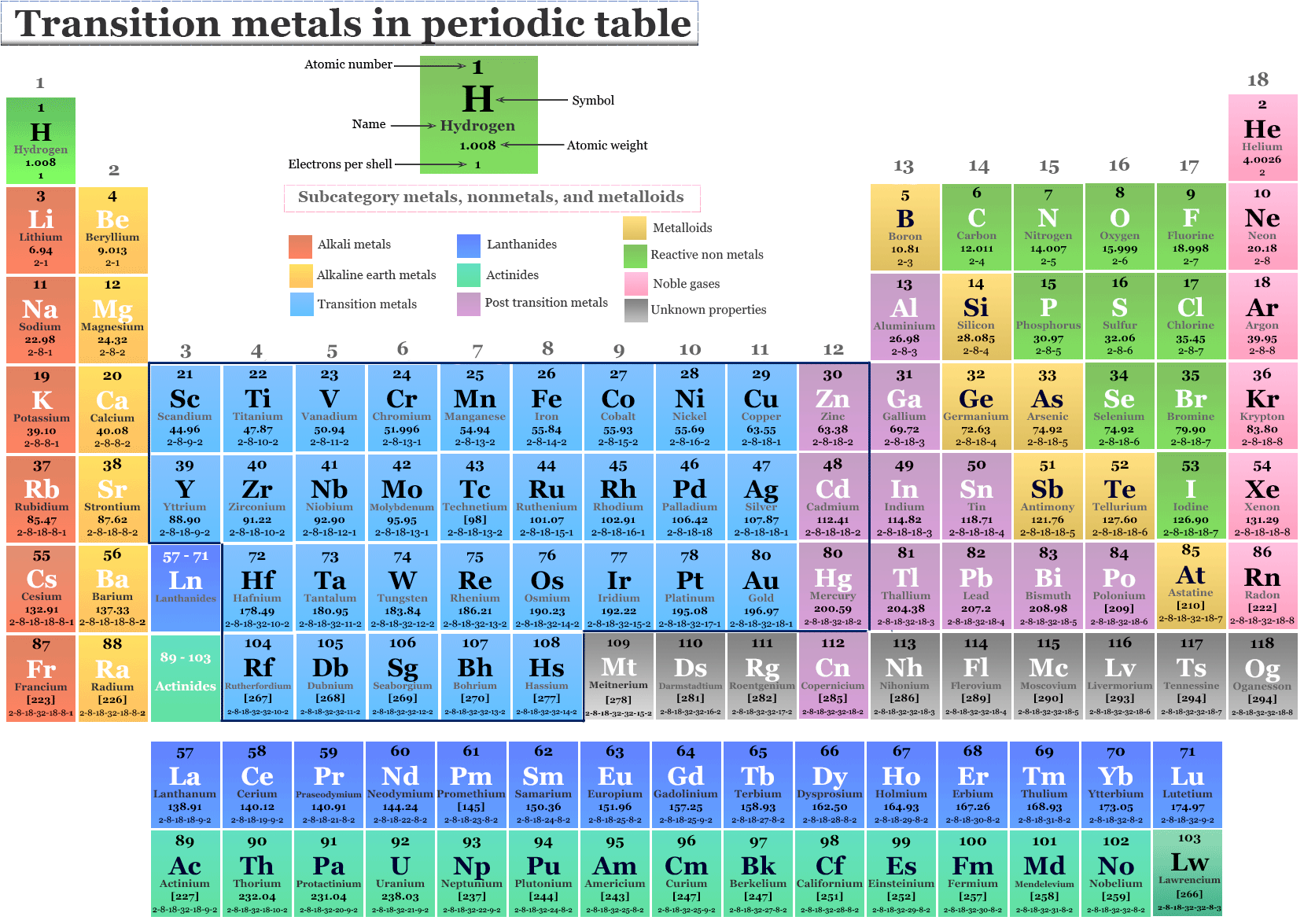
From building skyscrapers to creating life-saving medicines, these metals play a critical role. Their versatility makes them invaluable in our modern world.
Iron: The Backbone of Civilization
Iron (Fe) is arguably the most important Transition Metal. We use it to make steel, the material that holds up our buildings, bridges, and cars.
Without iron, the world as we know it would crumble (literally!). It's also crucial for carrying oxygen in our blood - talk about essential!
Plus, did you know that iron is the reason why Mars is red? The iron on its surface has oxidized, creating that rusty hue. Now that's a colorful contribution!
Copper: The Conductor of Electricity
Copper (Cu) is another superstar Transition Metal. Known for its excellent electrical conductivity, copper wires are found in nearly every electronic device.
Imagine a world without smartphones, computers, or the internet. Copper makes these technologies possible by efficiently transmitting electrical signals.
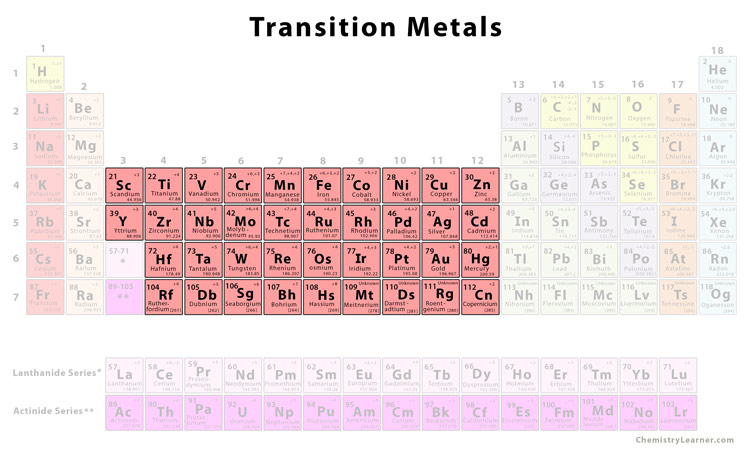
It’s also antimicrobial, making it great for door handles and other surfaces that people touch frequently. Copper is a real germ-fighting hero!
Gold and Silver: The Precious Metals
Ah, gold (Au) and silver (Ag), the glamorous duo! These Transition Metals have been prized for centuries for their beauty, rarity, and resistance to corrosion.
They're used in jewelry, coins, and even some electronics (gold is an excellent conductor, too!). Plus, they’re considered safe investments.
Ever wonder why Olympic medals are gold, silver, and bronze? It's a symbolic representation of excellence, with these metals representing the ultimate achievements.
Titanium: The Lightweight Champion
Titanium (Ti) is a Transition Metal known for its strength, light weight, and resistance to corrosion. It’s like the superhero of metals!
It's used in everything from airplanes and spacecraft to medical implants and sporting equipment. Talk about a versatile metal!
Its biocompatibility makes it ideal for implants, as the body doesn't reject it. It's literally helping people live better lives.
Zinc: The Protector Against Rust
Zinc (Zn) is another important Transition Metal, primarily known for its ability to protect other metals from corrosion. Think of it as the bodyguard of the metal world!
It's used in galvanization, a process where a layer of zinc is applied to steel to prevent rusting. This helps extend the lifespan of structures and equipment.
Zinc is also an essential nutrient for human health, playing a role in immune function and wound healing. It's not just protecting metal, it's protecting us too!
Chromium: The Colorful Protector
Chromium (Cr) is another corrosion fighter, known for its ability to make stainless steel "stainless." It also gives things a shiny, chrome finish!
It's used in car bumpers, kitchen appliances, and many other everyday items. Chromium adds a touch of sparkle and helps prevent rust.

Different oxidation states of chromium can produce a wide range of vibrant colors. These colors are used in pigments for paints and dyes. It’s like an artist in metal form!
Beyond the Basics
The world of Transition Metals is vast and complex. There are many other elements in this group, each with unique properties and applications.
From vanadium (V) to manganese (Mn) to nickel (Ni), they contribute to a wide array of technologies and industries. Exploring these metals can be a fascinating journey.
So, next time you encounter these metals in your daily life, remember their amazing properties and the crucial roles they play. They're more than just shiny objects. They're the backbone of modern society!
They’re Not Just Metals, They’re Superheroes
Ultimately, the Transition Metals are more than just a group of elements on the periodic table. They're the unsung heroes that make our modern world possible.
From the iron in our skyscrapers to the gold in our jewelry, these metals enrich our lives in countless ways. Their versatile personalities and impressive capabilities make them truly remarkable.
So, let's raise a glass (maybe made of stainless steel!) to the Transition Metals. They deserve our recognition and appreciation!
