What Are Alkali And Alkaline Earth Metals

Ever wonder what the heck scientists are rambling about when they mention alkali and alkaline earth metals? Don't worry, it's not as scary as it sounds! Think of them as the cool, reactive kids on the periodic table playground. Seriously, they're all about making connections and sometimes, things get a little...explosive. We'll break it down in a way that even your grandma who thinks chemistry is just mixing baking soda and vinegar will understand.
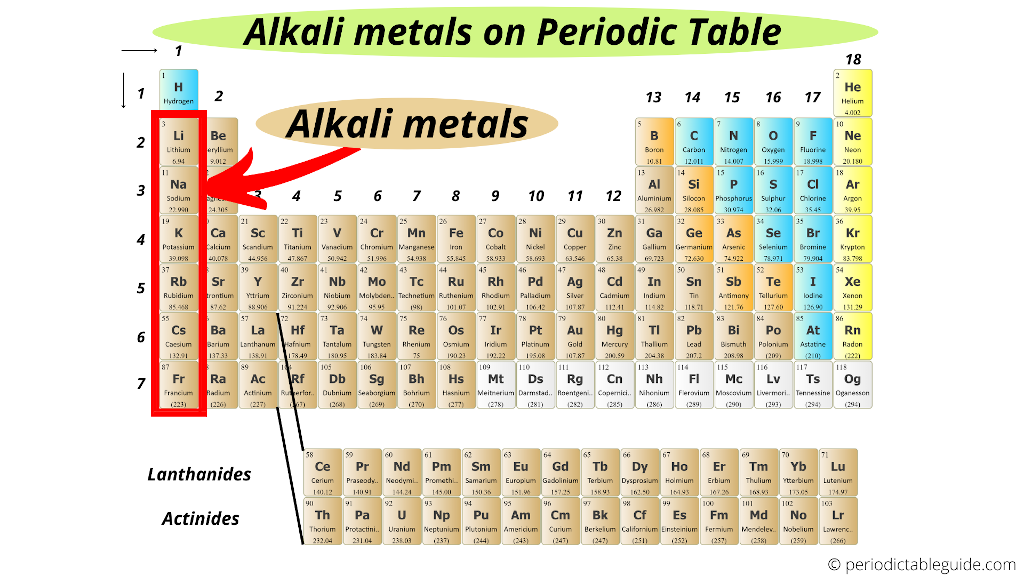
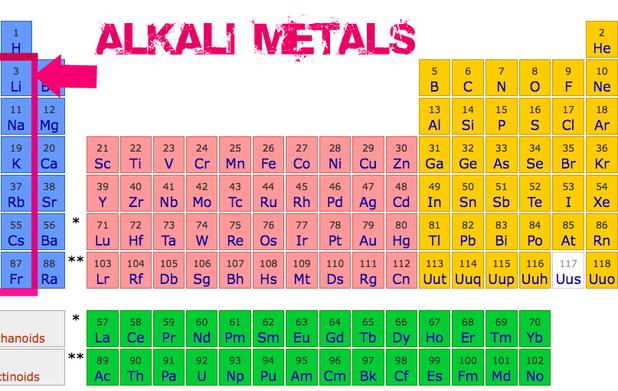
First, let's talk about the alkali metals. These guys are located in the first column of the periodic table – think of them as the front row, eager to be first at everything. We're talking lithium, sodium, potassium, rubidium, cesium, and francium. Now, imagine them as the overly enthusiastic people at a party. They are desperate to give something away, in this case, an electron. They’re so desperate, they'll react with just about anything to get rid of it!
Think of sodium (Na). You know, good old table salt (NaCl). Sodium all by itself? Throw it in water, and BOOM! It dances around, gets hot, and makes hydrogen gas. Don't try this at home, folks! It's way more exciting than watching paint dry, but also way more likely to end with a trip to the ER. My high school chemistry teacher, Mr. Abernathy, once did this demo, and a tiny piece of sodium jumped out of the beaker and almost hit the ceiling! We were all suitably impressed (and slightly terrified).
Must Read
Why are they so eager to react? Well, it all boils down to their electron configuration. They only have one lonely electron in their outer shell. They want to be stable, like the noble gases (those aloof, unreactive snobs on the far right of the periodic table). So, they ditch that electron as fast as they can to achieve that stable, full outer shell. It's like finally finding the perfect parking spot after circling the block for an hour.
Now, let's move on to the alkaline earth metals. These guys are in the second column of the periodic table: beryllium, magnesium, calcium, strontium, barium, and radium. They’re like the alkali metals’ slightly less hyperactive siblings. They also want to give away electrons, but they have two to give away instead of just one. So, they're a bit more…measured in their reactivity.
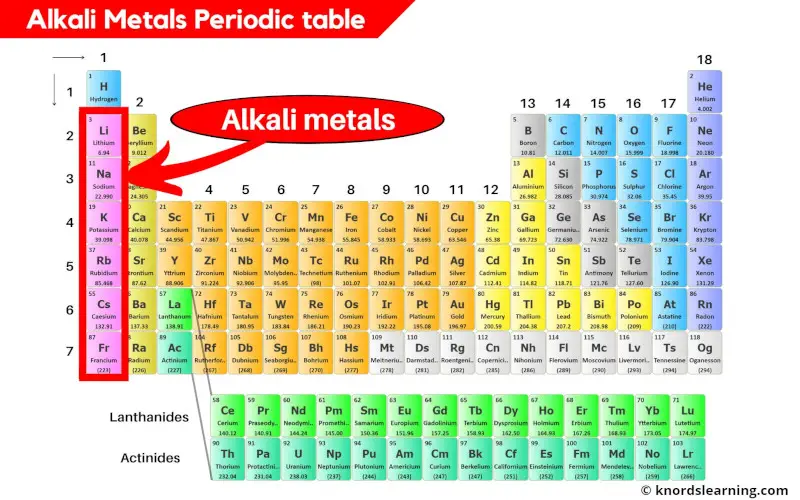
Think of them as the people at the party who are willing to share their snacks, but they're not going to shove them in your face. They need a little more convincing. But don’t get me wrong, they’re still reactive, just not quite as "hold my beer and watch this!" as the alkali metals.
Calcium (Ca) is a great example. You know, the stuff that makes your bones strong? You need it! Calcium reacts with water, but much more slowly than sodium. It's more of a slow simmer than a sudden explosion. You won't see it flying across the room (hopefully). Magnesium (Mg) is another important one, used in all sorts of things, from milk of magnesia to Epsom salts. It also burns with a brilliant white light – ever seen a sparkler? That's magnesium doing its thing!
So, what's the big takeaway?
The reactivity of both alkali and alkaline earth metals is all about their desire to lose electrons and achieve a stable electron configuration. The closer they are to the top of their respective columns, the more reactive they are. Francium and Radium are incredibly reactive (and radioactive!), so you're not likely to encounter them in your everyday life. Unless you're a nuclear physicist, in which case, you probably already knew all of this.
Think of it this way: The alkali metals are like that friend who always volunteers for everything, even if it's a terrible idea. The alkaline earth metals are like the friend who's willing to help, but they want to know what they're getting into first. Both are essential in their own way, just like these metals are essential in chemistry and, surprisingly, in our everyday lives.
From making table salt to keeping our bones strong, alkali and alkaline earth metals play a vital role in the world around us. They might be a little reactive and a little crazy, but without them, life would be a whole lot less interesting (and a whole lot less salty).