What Acid Is In Lead Acid Batteries
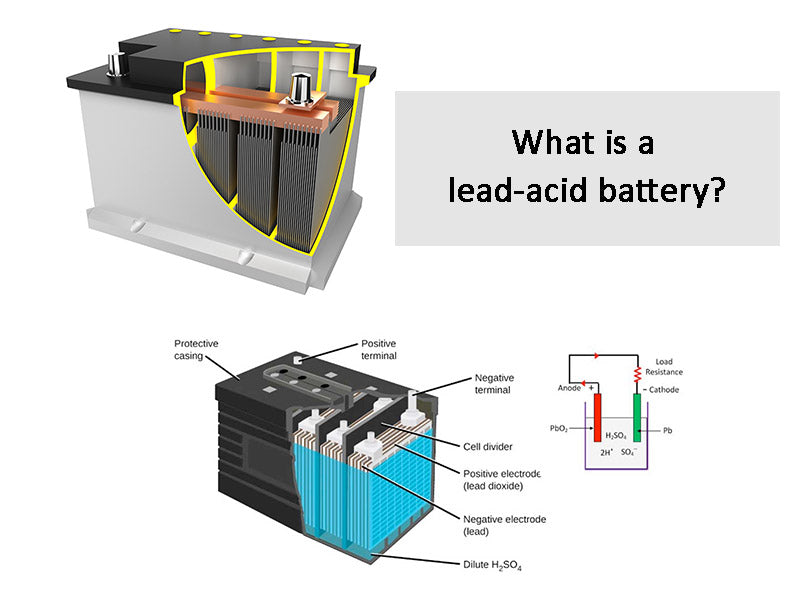
Okay, let's talk about something surprisingly...artistic. Something you probably associate more with your car than a canvas: lead-acid batteries! Before you imagine us advocating dismantling car batteries (don't do that!), let's clarify. We're not talking about battery acid in the artistic sense. We're here to uncover a little chemistry secret: what is the acid in those ubiquitous batteries?
The star of the show is sulfuric acid. Yes, that sounds intimidating! But understanding its role is key to appreciating the clever engineering of lead-acid batteries, which, in turn, can spark some interesting thought experiments for artists, hobbyists, and anyone curious about the world around them.
Why should this interest an artist or hobbyist? Well, consider the controlled chemical reaction happening within the battery. It's a miniature, self-contained power source. This concept can be inspiring for kinetic art, automated sculptures, or even just understanding the flow of energy in a system. Think about it: an artist could create a piece where the depletion of a small, safe battery (not a car battery!) triggers a series of events, like a miniature Rube Goldberg machine that paints its own abstract artwork. A budding electrical engineer might use this knowledge to create miniature power circuits to drive tiny robots. The possibilities, fueled by a basic understanding of sulfuric acid's role, are surprisingly diverse!
Must Read
Imagine a sculptor using tiny lead-acid batteries (designed for electronics, of course!) to power small lights illuminating specific aspects of their work. Or a textile artist integrating conductive threads powered by a miniature battery to create interactive clothing. The subjects could range from the purely abstract – pulsating colors and shifting patterns – to the representational, such as a scale model of a city brought to life with glowing windows and moving parts.
Now, for a word of serious caution: we absolutely do not recommend playing with sulfuric acid from car batteries. It's extremely corrosive and dangerous. However, learning about electrochemistry kits designed for educational purposes can be a safe alternative. These kits often use diluted sulfuric acid or other electrolytes to demonstrate battery principles. You can also explore online simulations and virtual labs to experiment with battery designs without any risk.

Instead of handling dangerous chemicals, consider exploring the concept of the electrochemical reaction. Study diagrams of lead-acid batteries and understand how the sulfuric acid interacts with the lead plates to generate electricity. This knowledge can be surprisingly empowering, allowing you to understand the technology that powers so much of our modern world.
The enjoyment comes from the aha! moment of understanding how seemingly complex devices work. It's about connecting the dots between chemistry, engineering, and creative expression. It's about recognizing the potential for innovation and artistic exploration hidden within even the most mundane objects. So, while you might not be pouring sulfuric acid on a canvas anytime soon, understanding its role in a lead-acid battery can open up a whole new world of artistic and intellectual possibilities. And that’s pretty darn enjoyable.



