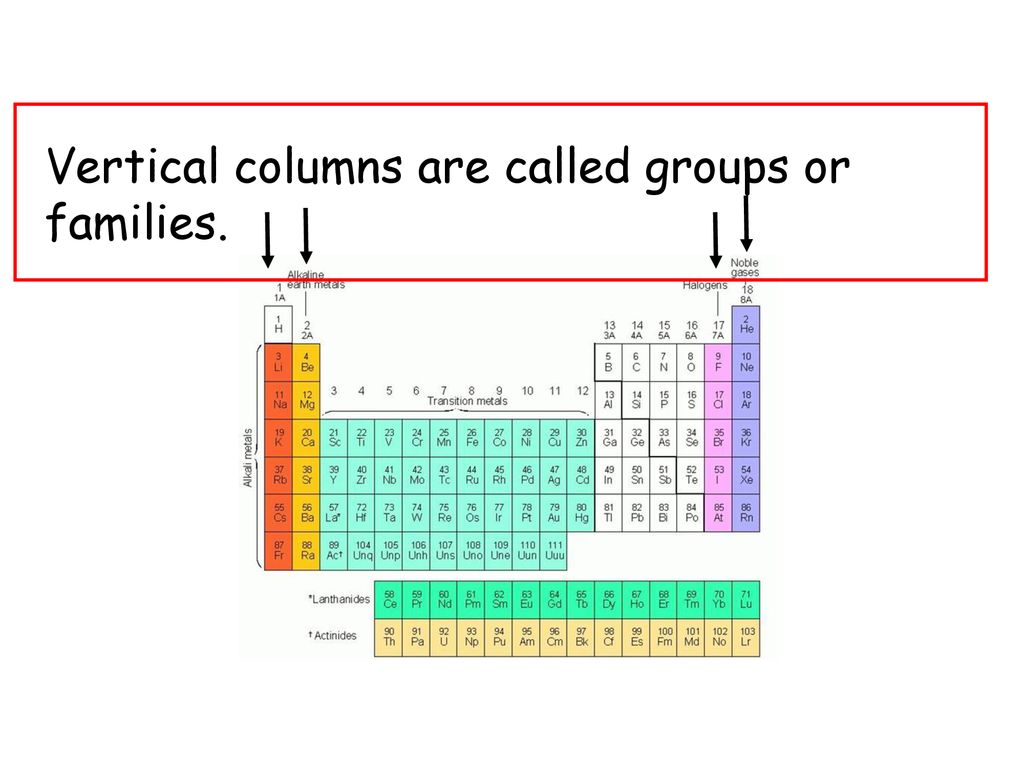
Vertical Columns On The Periodic Table Are Called
+columns+of+the+periodic+table+(there+are+18)+are+called+groups+or+families..jpg)
Hey there, science buddy! Ever looked at the periodic table and thought, "Wow, that's a lot of squares?" I know I have! It's like a giant, organized puzzle of all the elements that make up, well, everything! But have you ever wondered about those vertical columns? You know, the ones that go up and down like elevator shafts for tiny atoms?
Well, guess what? They have a name! And it's a pretty important one in the world of chemistry. Drumroll please…
They're called Groups!
Yep, that's it! Groups. Simple, right? I mean, they could have called them "Super Atomic Squads" or "Vertical Element Ventures," but I guess "Groups" gets the job done. (Although, I kind of like "Super Atomic Squads"… maybe we should start a petition?).
Must Read
So, why are these Groups so special anyway? Is it just because they look nice all lined up? Nope! There's a much more important reason than aesthetics. The elements in the same Group share similar chemical properties.
Think of it like this: Imagine you're putting together a sports team. You want players who can work well together, right? Elements in the same Group are like teammates. They have similar "skills" (or, in chemistry speak, they react in similar ways).

For example, the elements in Group 1, also known as the alkali metals (like sodium and potassium), are all super reactive! They're so eager to bond with other elements that they're never found in their pure form in nature. They're like the overly-enthusiastic players who jump for every ball, even if it's heading out of bounds! (Someone needs to tell them to chill, but you gotta love the energy!)
Then you have Group 17, the halogens (like chlorine and fluorine). These elements are also very reactive, but in a different way. They're like the players who are always trying to steal the ball from the other team. They're just really good at attracting electrons.
And then there's Group 18, the noble gases (like helium and neon). These are the chill dudes on the team. They're so stable and happy with their electron arrangement that they rarely react with anyone! They're like the zen masters of the periodic table, perfectly content with themselves.
So, as you can see, knowing which Group an element belongs to can tell you a lot about its behavior. It's like having a cheat sheet for chemistry! No more guessing whether an element will explode on contact with water (looking at you, sodium!). You can just check its Group and know what to expect.
But wait, there's more! The number of valence electrons (those important electrons in the outermost shell) is the same for all elements in a Group. And guess what dictates how an atom reacts? That’s right, the number of valence electrons!

Basically, all this sharing of similar properties within a Group happens because of the number of electrons in the outermost shell. Isn’t that just mind-blowing?
Now, I know the periodic table can seem intimidating at first, but trust me, it's actually pretty awesome! It's like a map of the universe, showing you all the building blocks of everything around you. And understanding what those Groups mean is a huge step toward unlocking the secrets of chemistry!
So the next time you see a vertical column on the periodic table, remember that it's not just a random line-up of elements. It's a Group of elements with shared characteristics, and it's waiting to reveal its secrets to you. Go forth and explore the wonders of the periodic table! You've got this!