Van T Hoff Factor Calculator
Hey there, science buddy! Ever felt like you're stirring a drink and something just... doesn't dissolve quite the way you expected? Like you put in one spoon of sugar, but it acts like you put in, I don't know, one-and-a-half? That, my friend, is where the Van 't Hoff factor waltzes onto the stage!
Now, I know what you might be thinking: "Van 't Hoff? Sounds like a villain from a bad spy movie!" But trust me, it's much more interesting (and less likely to try and take over the world). It's actually named after a Dutch physical chemist, Jacobus Henricus van 't Hoff, and it helps us understand how substances behave when they dissolve in liquids.
So, What Exactly Is the Van 't Hoff Factor?
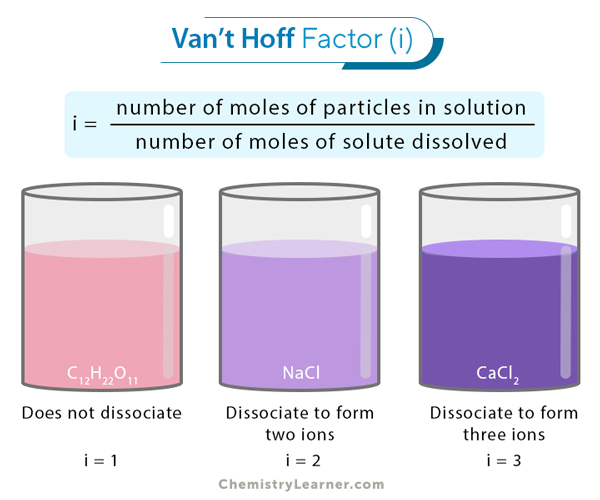
In a nutshell, the Van 't Hoff factor (often represented by the letter i) tells you how many particles one formula unit of a substance will split into when it dissolves. Think of it as the "dissociation multiplier."
Must Read
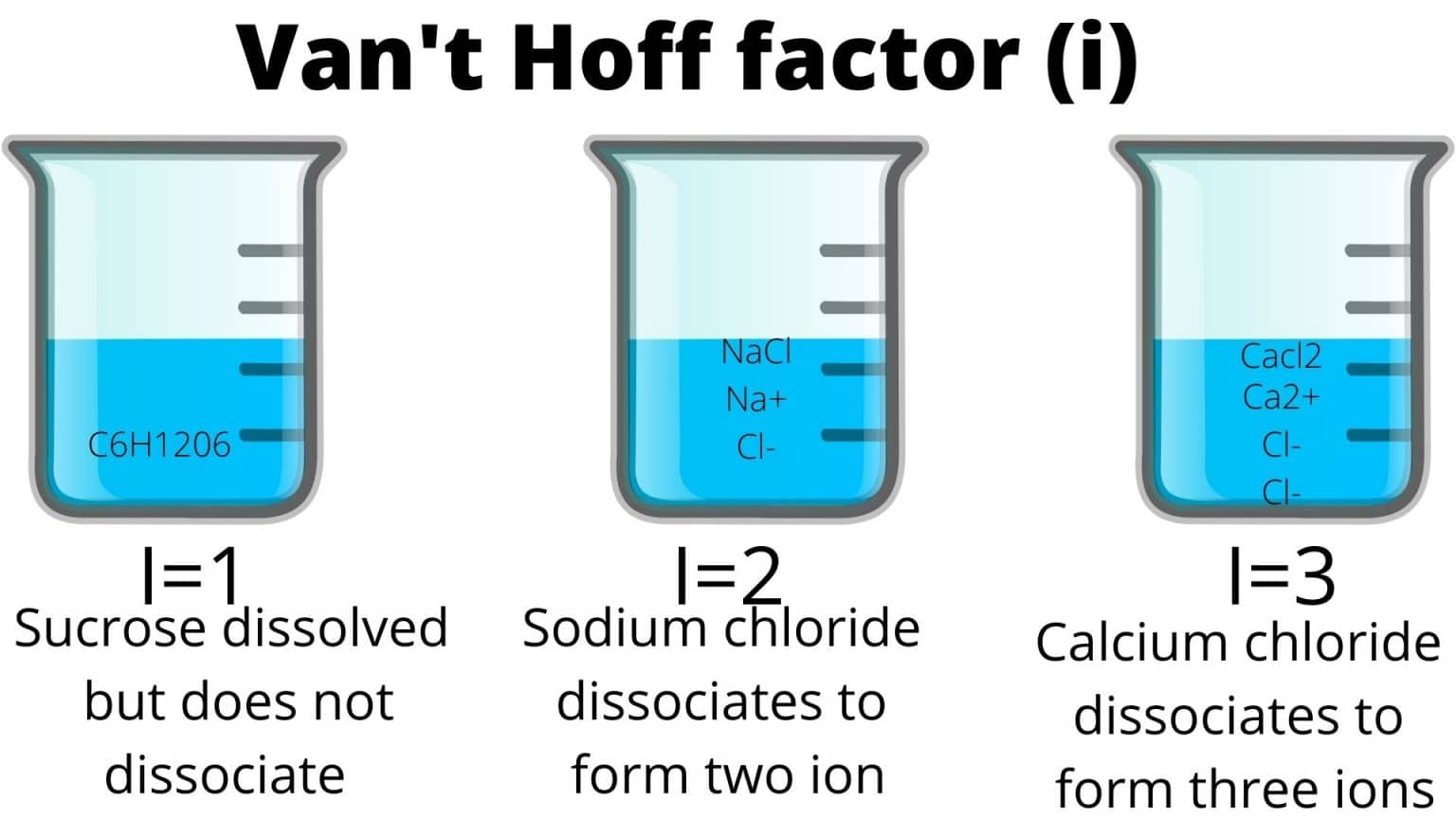
For example, sugar (like sucrose, C12H22O11) doesn't break apart when it dissolves. It stays as one single sugar molecule floating around. So, its Van 't Hoff factor is 1. One sugar molecule in, one sugar molecule floating about. Simple, right?
But things get a bit more exciting with ionic compounds, like good ol' table salt (NaCl). When salt dissolves in water, it breaks down into sodium ions (Na+) and chloride ions (Cl-). So, one NaCl unit becomes two particles. Therefore, its Van 't Hoff factor is (theoretically!) 2. Notice that “theoretically!”, that’s a little secret for later.

And guess what? Things can get even wilder! Calcium chloride (CaCl2) dissociates into one calcium ion (Ca2+) and two chloride ions (Cl-). That's a total of three particles, giving it a Van 't Hoff factor of 3. Boom! Party of three in your solution!
Why Do We Need to Know This?
Okay, so besides impressing your friends at your next chemistry-themed party (guaranteed to be a hit!), the Van 't Hoff factor is super useful for calculating things like:

- Osmotic pressure: The pressure required to stop osmosis (the movement of water across a semipermeable membrane). Think of it as the pressure needed to stop your cells from exploding (or shriveling up!).
- Freezing point depression: How much lower a solution's freezing point is compared to the pure solvent (like water). This is why we use salt on icy roads! (And why your ice cream melts faster when you add salt – sorry!).
- Boiling point elevation: How much higher a solution's boiling point is compared to the pure solvent. Knowing this helps you cook your pasta perfectly! (Maybe. Okay, probably not, but it's still cool!).
Enter: The Van 't Hoff Factor Calculator (Your New Best Friend)
Now, while understanding the concept is crucial, sometimes you just need to plug in some numbers and get an answer. That's where a Van 't Hoff factor calculator comes in! These online tools (and sometimes even built into your trusty calculator) can help you quickly determine the Van 't Hoff factor based on the compound you're working with.
Generally, you'll need to know the chemical formula of the compound. Then, the calculator will (usually) make some assumptions about complete dissociation to give you a theoretical value. But remember that "theoretically" from earlier? Here's the deal...
The "Real World" Twist: It's Not Always Perfect
In the real world, things are never quite as tidy as our theoretical models predict. Sometimes, ionic compounds don't completely dissociate. Some ions might stick together a little bit, forming ion pairs or larger clusters. This means the actual Van 't Hoff factor might be slightly lower than the theoretical value.
Factors like concentration and temperature can also affect the Van 't Hoff factor. In highly concentrated solutions, there's a greater chance of ions interacting with each other, reducing the effective number of particles.

So, when you use a Van 't Hoff factor calculator, keep in mind that it's often giving you an approximation. For more precise calculations, you might need to consider experimental data or more advanced models.
In Conclusion: Go Forth and Dissolve!
The Van 't Hoff factor might sound intimidating at first, but it's really just a tool to help us understand the fascinating world of solutions and their properties. Whether you're a student, a scientist, or just someone curious about how things work, understanding this concept can give you a deeper appreciation for the chemistry all around us.
So, go ahead, embrace the Van 't Hoff factor! Experiment with different solutes and solvents. Play around with a calculator. And remember, even if things don't always dissolve quite as expected, that's just science being science – full of surprises and opportunities to learn something new! Now, go forth and dissolve the world!
