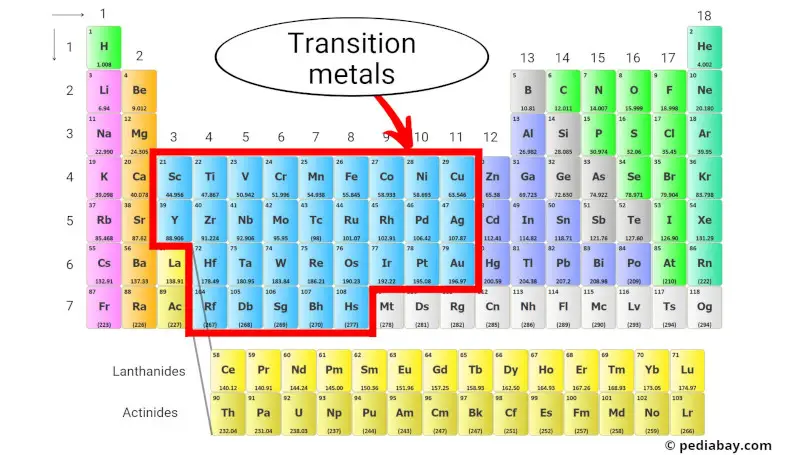
Transition Metals On A Periodic Table

Ever wondered what makes a dazzling piece of jewelry shine or what's inside the wires that power your phone? The answer, in many cases, lies with a fascinating group of elements on the periodic table: the transition metals! These elements aren't just science textbook fodder; they're the workhorses of our modern world, making them surprisingly fun and incredibly useful to understand, even on a basic level.
Why bother learning about transition metals? Well, for beginners, they offer a gateway into understanding how the periodic table is organized and how different elements behave. Spotting them on the table and understanding they often have colorful compounds (think vibrant blue copper sulfate crystals) is a great first step. For families, exploring transition metals can be a fun, hands-on learning experience. You can point out examples in everyday objects – from the iron in your pots and pans to the titanium in sports equipment. It’s a chance to connect abstract science concepts to the real world! And for hobbyists like artists, jewelry makers, or even gardeners, knowing about transition metals can be incredibly valuable. Understanding their properties can inform your choices of materials and even impact your creative processes.
So, what exactly are transition metals? They’re the elements found in the middle block of the periodic table, generally groups 3 to 12. Think of them as the busy bees of the element world, constantly forming bonds and creating interesting compounds. A key characteristic is their ability to form multiple oxidation states, meaning they can lose different numbers of electrons, leading to that vibrant array of colors we mentioned earlier. For instance, iron (Fe) can be found as rust (iron oxide), or in hemoglobin, the protein in our blood that carries oxygen. Copper (Cu) is vital for electrical wiring, and silver (Ag) is prized for its conductivity and beauty in jewelry. Gold (Au), platinum (Pt), and palladium (Pd) are other well-known examples, valued for their resistance to corrosion and unique chemical properties.
Must Read
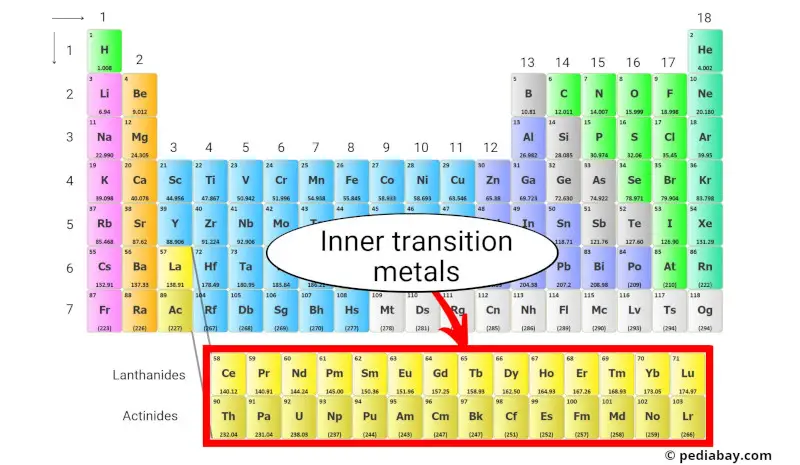
There are also some interesting variations within the transition metal family. The lanthanides and actinides, often listed separately at the bottom of the periodic table, are technically part of the transition metal group, but have their own special properties. These elements, including uranium and plutonium, are particularly relevant to nuclear chemistry and energy.
Ready to dive in? Here are a few simple tips to get started:
- Explore a periodic table online: Many interactive periodic tables online provide information about each element, including its uses and properties.
- Look around you: Identify objects containing transition metals. Research what that metal contributes to the object's function.
- Conduct simple experiments (with adult supervision!): Growing copper sulfate crystals is a classic and engaging project.
Learning about transition metals doesn't have to be daunting. By understanding their basic properties and recognizing their presence in everyday life, you can unlock a deeper appreciation for the science that shapes our world. So, embrace the curiosity and enjoy the colorful journey into the realm of transition metals!