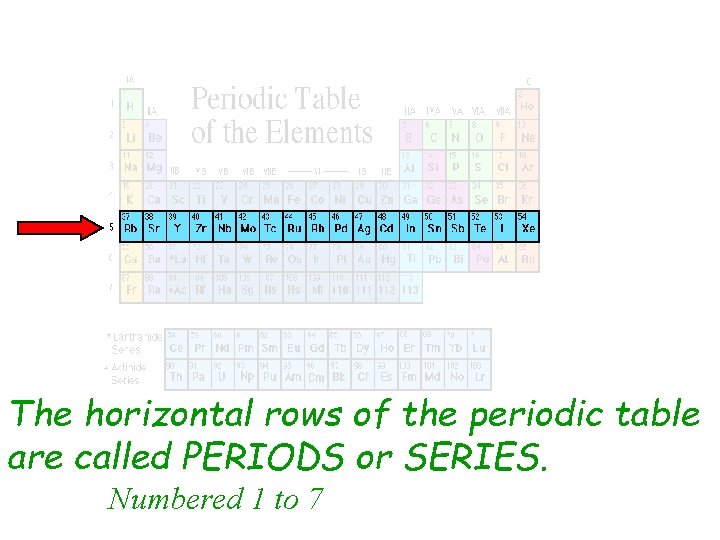
The Horizontal Rows On The Periodic Table Are Called
Ever stared at the periodic table and felt like you were looking at some kind of alien city grid? I know I have! All those squares, those weird abbreviations... it's enough to make anyone's head spin. But don't worry, we're here to demystify one little piece of that puzzle: the horizontal rows.
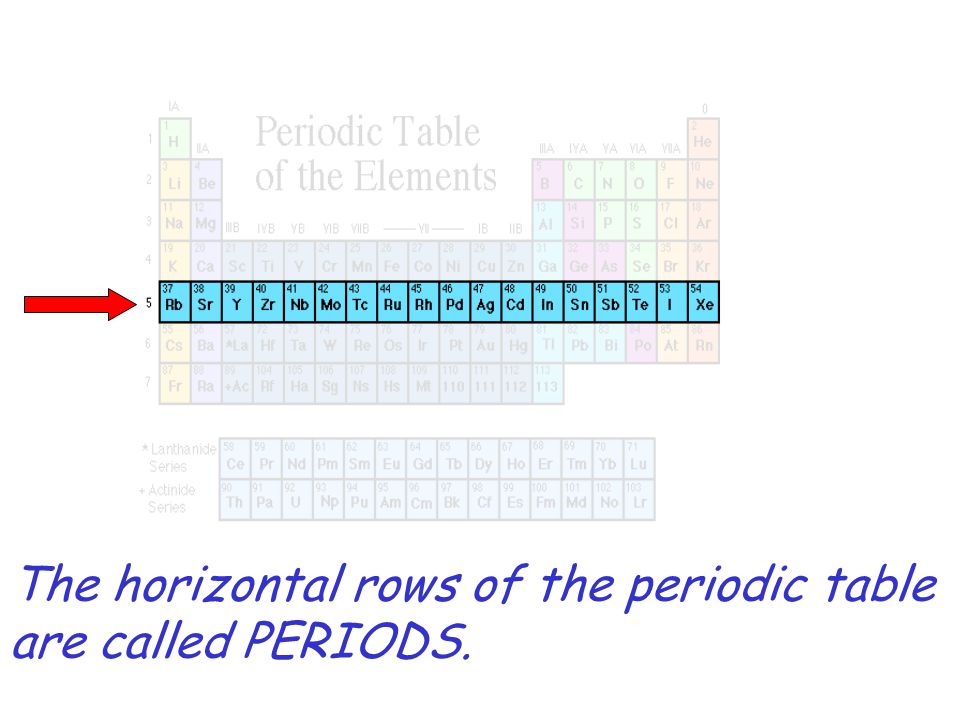
Think of the periodic table like a seating chart at a really, really weird dinner party. You've got all these elements, right? And they're all lined up in rows and columns. Now, we're not talking about the vertical columns (those are called groups or families, and that's a story for another time!). We're focusing on the horizontal rows.
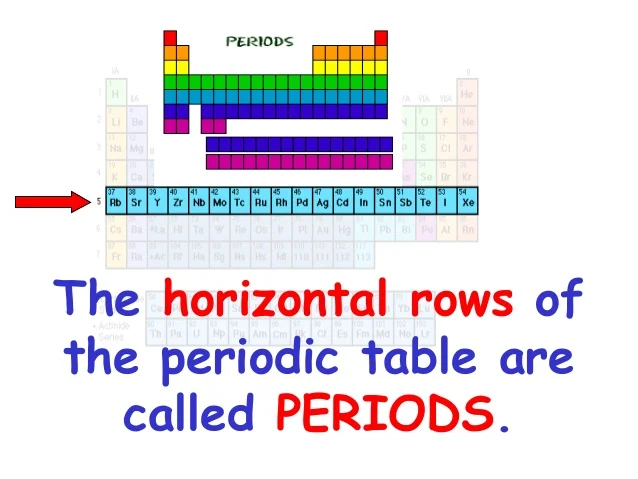
So, what are these rows called? Drumroll please... they're called periods.
Must Read
Yep, periods. Not that kind of period. We’re talking chemistry here, folks. Though, let's be honest, sometimes chemistry can feel just as unpredictable as… well, you know.
Periods: Not Just a Punctuation Mark
I know, I know, the name "period" doesn't exactly scream "excitement." It sounds more like something you'd find at the end of a sentence, not something that holds valuable information about the building blocks of the universe! But stick with me, because these periods are actually pretty darn cool.

Each period represents a new energy level for the electrons swirling around the nucleus of an atom. Imagine it like the rings of an onion, or maybe the levels of a parking garage. As you go down the periodic table, each new period adds another "ring" or "level" to the atom's electron structure.
Think of it like this: the first period (Hydrogen and Helium) is like a tiny studio apartment. Just enough room for the bare essentials. But as you move down to the second period (Lithium to Neon), you’re upgrading to a one-bedroom apartment – more space for more electrons! And by the time you get to the seventh period (Francium to Oganesson), you're practically living in a mansion with enough electron rooms to make your head spin. Okay, maybe not literally a mansion, but you get the idea.
What Makes a Period a Period?
So, what determines which element ends up in which period? Well, it all comes down to the number of electron shells (those energy levels we were talking about). Elements in the same period have the same number of electron shells. As you move across a period from left to right, the number of protons and electrons increases. This affects the atom's properties, like its electronegativity (how much it wants to grab onto electrons) and its ionization energy (how much energy it takes to rip off an electron).

In other words, elements in the same period have similar, but gradually changing, properties. It’s like a family with siblings – they share certain traits, but they're also individuals with their own quirks and personalities.
A Few Fun Facts (Because Why Not?)
There are seven periods in the periodic table that we know about right now. While scientists continue to research and synthesize new elements, the current periodic table ends with the seventh period.
The elements in the first period are super special because they only have one electron shell. It's like they're the minimalist Marie Kondo-approved elements of the periodic table!
The length of each period varies. Some periods are short (like the first period with just two elements), while others are much longer (like the sixth and seventh periods which include the lanthanides and actinides – those guys hanging out at the bottom of the table like they're in timeout).
So, the next time you see a periodic table, don't just see a confusing grid. Remember those horizontal rows, the periods, and the story they tell about electron shells and changing properties. It's like unlocking a secret code to understanding the universe, one element at a time. And who knows? Maybe you'll even impress your friends at your next dinner party with your newfound knowledge! Just try not to talk about electronegativity too much, or you might clear the room.