Sulfur Lewis Dot Structure

Hey there, chemistry curious folks! Ever stared at a chemical formula and felt a wave of confusion wash over you? Yeah, me too. But trust me, things get a whole lot clearer (and dare I say, kinda fun?) when you start visualizing things with Lewis Dot Structures. And today, we're diving headfirst into one of my favorites: Sulfur!
So, what is a Lewis Dot Structure anyway? Think of it as a chemical shorthand, a visual way to represent the valence electrons of an atom – those crucial outer-shell electrons that are responsible for all the bonding action. Imagine them as the atom's social butterflies, always ready to mingle and form connections.
Why Bother with Lewis Dot Structures?
Good question! Why spend time drawing dots when you could be watching cat videos? Well, Lewis Dot Structures give you a sneak peek into how an atom will likely interact with others. They help us predict what kind of bonds will form (like those cute covalent bonds where electrons are shared!), and ultimately, what the molecule will look like. It's like having a cheat sheet to understanding the chemical world!
Must Read
Think of it this way: if atoms were people, valence electrons would be their personalities. Understanding those "personalities" helps you understand how they'll get along with others. Makes sense, right?
Sulfur: The Many-Faced Atom
Okay, let's get down to business. Sulfur, represented by the symbol 'S', is a super interesting element. Why? Because it's kinda a chameleon! It can form all sorts of different compounds and take on different oxidation states, which basically means it can share varying numbers of its electrons. It's like the cool kid who can hang out with any group.

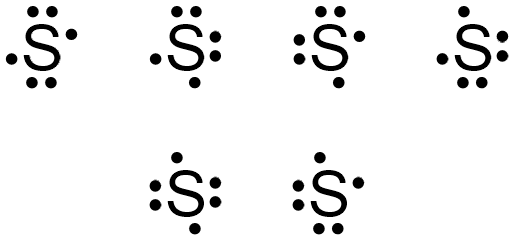
Sulfur sits smack-dab in Group 16 (also known as the chalcogens) on the periodic table. This is super important because it tells us something crucial: Sulfur has 6 valence electrons. Remember, the group number often corresponds to the number of valence electrons (with a few exceptions, of course – chemistry loves its exceptions!).
Drawing the Sulfur Lewis Dot Structure: Dot by Dot
Alright, grab your imaginary pencils (or, you know, use a real one if you're feeling fancy) and let's draw this thing. Here's the step-by-step:
- Write the Symbol: First, write down the chemical symbol for sulfur: 'S'. Simple enough, yeah?
- Add the Dots: Now, we need to represent those 6 valence electrons. Think of the symbol 'S' as being surrounded by four "slots" (top, bottom, left, and right). We're going to fill those slots with dots, representing electrons.
- Pair 'Em Up (or Not!): Start by placing one dot in each slot. So, one at the top, one at the bottom, one on the left, and one on the right. That's four dots down, two to go. Now, pair up the remaining dots with any of the slots. It doesn't really matter which ones you choose!
Voila! You should now have a sulfur atom with six dots around it. Two of those dots are paired up (representing lone pairs of electrons – they're not actively bonding with anything), and two are single (unpaired). Those unpaired electrons are the key players in sulfur's bonding shenanigans!

Sulfur's Bonding Potential: Why It's So Versatile
Those unpaired electrons are super important. They're ready and willing to form bonds with other atoms. This explains why sulfur can form two single bonds (think H2S, hydrogen sulfide – smells like rotten eggs, by the way!). It can also form one double bond (think CS2, carbon disulfide). Or even a combination of single and double bonds, if the situation calls for it! Talk about adaptable!
Why is this so important? Well, think about all the different compounds that contain sulfur: proteins (sulfur-containing amino acids help them fold correctly!), sulfuric acid (a super important industrial chemical), and even some antibiotics. Sulfur's ability to form various bonds makes it a crucial player in a wide range of chemical processes.

More Than Just Dots: A Deeper Understanding
While Lewis Dot Structures are simplified representations, they offer a powerful tool for understanding chemical bonding. They help us visualize how atoms share electrons and predict the shapes of molecules. It’s like having a roadmap to the molecular world!
So, next time you see a chemical formula, don't be intimidated. Remember sulfur and its friendly dots! Take a deep breath, draw out the Lewis Dot Structure, and unlock the secrets of the chemical world. You might just surprise yourself with what you discover!
Who knew dots could be so darn insightful, right?