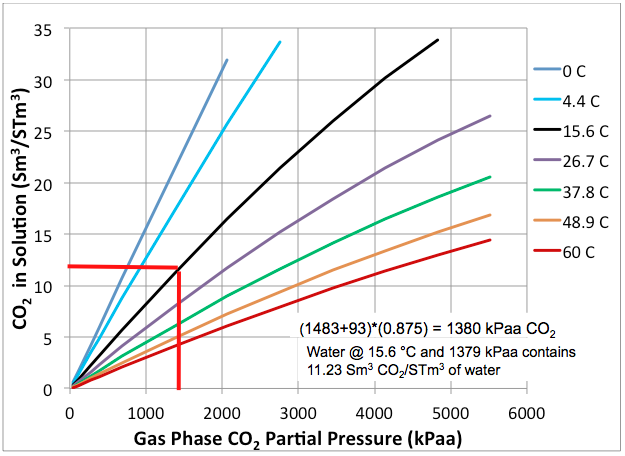
Solubility Of Co2 In Water

Ever poured yourself a nice, fizzy drink? That glorious sound, those tiny bubbles dancing upwards? Pure bliss! It’s a moment of sparkling perfection. But then, too quickly, it just... goes flat. That lively fizz vanishes. It becomes just a sad, quiet liquid. What happened? Who’s the culprit? I’m here to share my slightly "unpopular" opinion.
Meet our main character: Carbon Dioxide, or CO2 for short. This invisible gas is the secret behind all those wonderful bubbles. It’s what makes soda, sparkling water, and even beer so refreshing. Without CO2, these drinks would be pretty dull, wouldn't they?
The "Relationship" with Water
When we talk about solubility, we're basically asking: how well does one thing dissolve into another? Can water actually hold onto our bubbly friend, CO2? The answer is a bit like a complicated relationship status on social media. Water can hold CO2, but it’s often a very temporary arrangement.
Must Read
My take? CO2 doesn't truly love water. It tolerates it, at best. It's like that guest at a party who's polite, but you can tell they’re constantly checking their watch, plotting their grand exit. Water tries its best to keep CO2 around, but our gaseous friend is always looking for an escape route. It's not really committed to the liquid life.
The Great Escape Factors
Ever noticed how a warm can of soda goes flat almost instantly? Meanwhile, a super cold one keeps its fizz for ages. This isn't magic! It’s all about temperature. When water gets warmer, the tiny water molecules move faster and get all jiggly. This makes it much easier for the dissolved CO2 molecules to break free and float away.
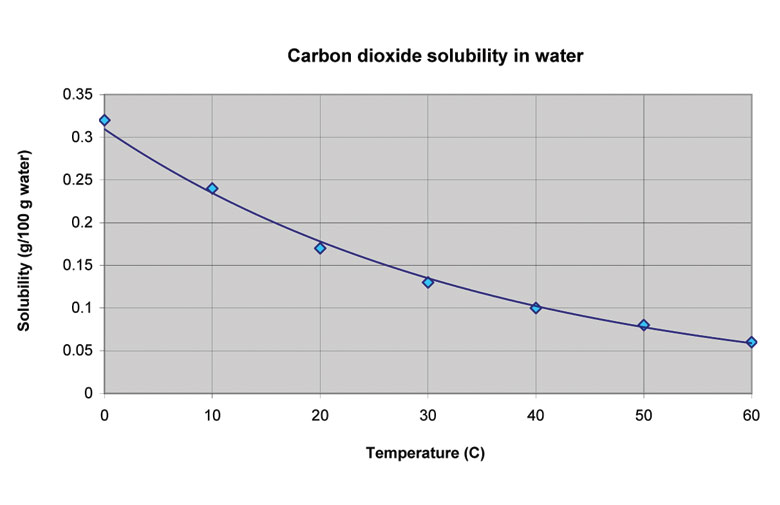
Warm water is practically a CO2 eviction notice.
It’s like someone turning up the heat at that party. Everyone gets uncomfortable, and the guests start leaving! Colder water, however, is a much better host. The molecules are slower, allowing water to hug onto the CO2 molecules more tightly.
Then there's pressure. Think about a sealed soda bottle or can. There's a lot of pressure inside that container. This high pressure literally forces the CO2 to stay dissolved in the water. It’s like a bouncer at a club, keeping everyone inside. The moment you twist off the cap or pull the tab, you hear that satisfying hiss. That’s the sound of the pressure being released!
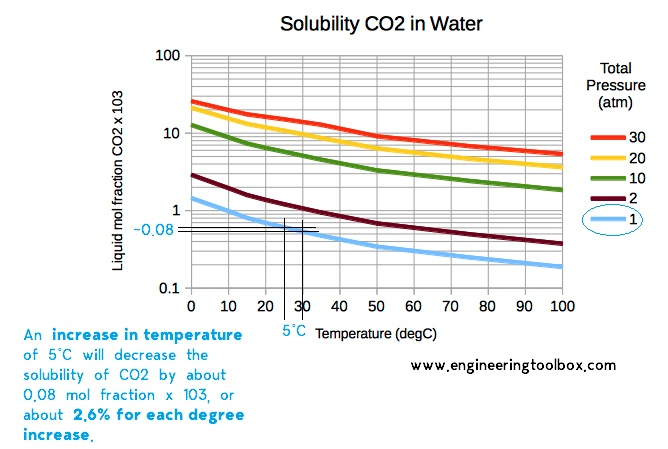
With the bouncer (pressure) gone, CO2 sees its chance. It makes a mad dash for freedom! Those tiny bubbles that suddenly appear and rise to the surface? Those are little clusters of CO2 finally making their grand escape from the water. They're like miniature submarines, surfacing and then vanishing into the air. They’ve successfully pulled off their escape plan.
The Unavoidable Truth
So, the next time your fizzy drink disappoints you by losing its sparkle, don't blame yourself. Don't even blame the drink's quality. Blame CO2. It's a fickle friend, always looking for a way out. Its solubility in water is a temporary truce, not a lifelong commitment. We just have to accept that our bubbly joy is often fleeting. Perhaps the real lesson is: enjoy your fizz while it lasts. And maybe, just maybe, drink it a little faster next time!