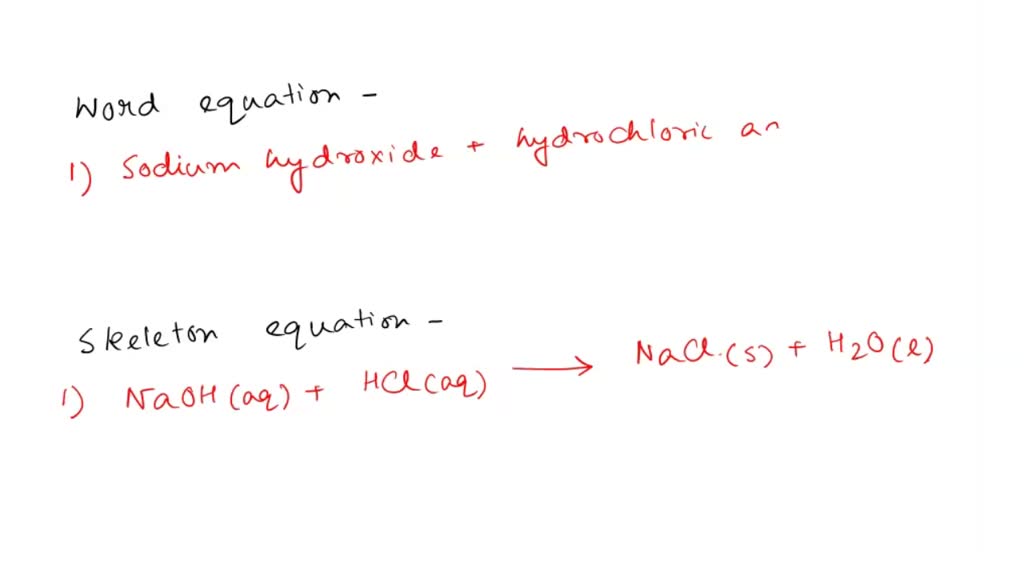
Sodium Hydroxide And Hydrochloric Acid

Okay, let's talk about something that sounds intimidating but is actually just like that quirky couple you know – Sodium Hydroxide and Hydrochloric Acid. Individually, they're a bit intense, but put them together, and they create something totally normal (well, mostly!). Think of it like that friend who's super into extreme sports and another who only reads philosophy – weird mix, but somehow, they make it work.
Sodium Hydroxide: The Drano of Chemistry
Sodium Hydroxide (NaOH), also known as lye or caustic soda, is the stuff that can unclog your drains. Imagine it as the chemistry world's version of a tiny, relentless plumber. It's a strong base, which means it’s basically the opposite of an acid. Think of it as the grumpy old man of chemicals; it reacts strongly with pretty much anything acidic it comes across. It’s used in everything from making soap (that clean feeling? Thank Sodium Hydroxide!) to paper production.
Just like you wouldn’t want your grumpy uncle handling your delicate china, you wouldn’t want to mess around with concentrated Sodium Hydroxide without protection. It can cause some serious burns. Remember that time you accidentally touched a hot pan? Yeah, it's a bit like that, but chemical. So, always wear gloves and eye protection! Safety first, folks!
Must Read
Hydrochloric Acid: The Stomach’s Secret Weapon
Hydrochloric Acid (HCl) is a strong acid and is what your stomach uses to break down food. Ever felt that heartburn sensation after a huge meal? That's a bit of stomach acid escaping, reminding you just how powerful this stuff is. It’s like your stomach has its own little chemistry lab going on, breaking down that triple cheeseburger you probably shouldn’t have eaten.
Beyond digestion, Hydrochloric Acid is used in industry for cleaning metal, etching, and even in the production of gelatin. It's a versatile chemical, a bit like that Swiss Army knife you always forget you have until you desperately need it. But just like you wouldn't use that knife to perform open-heart surgery, you also need to be careful with Hydrochloric Acid.
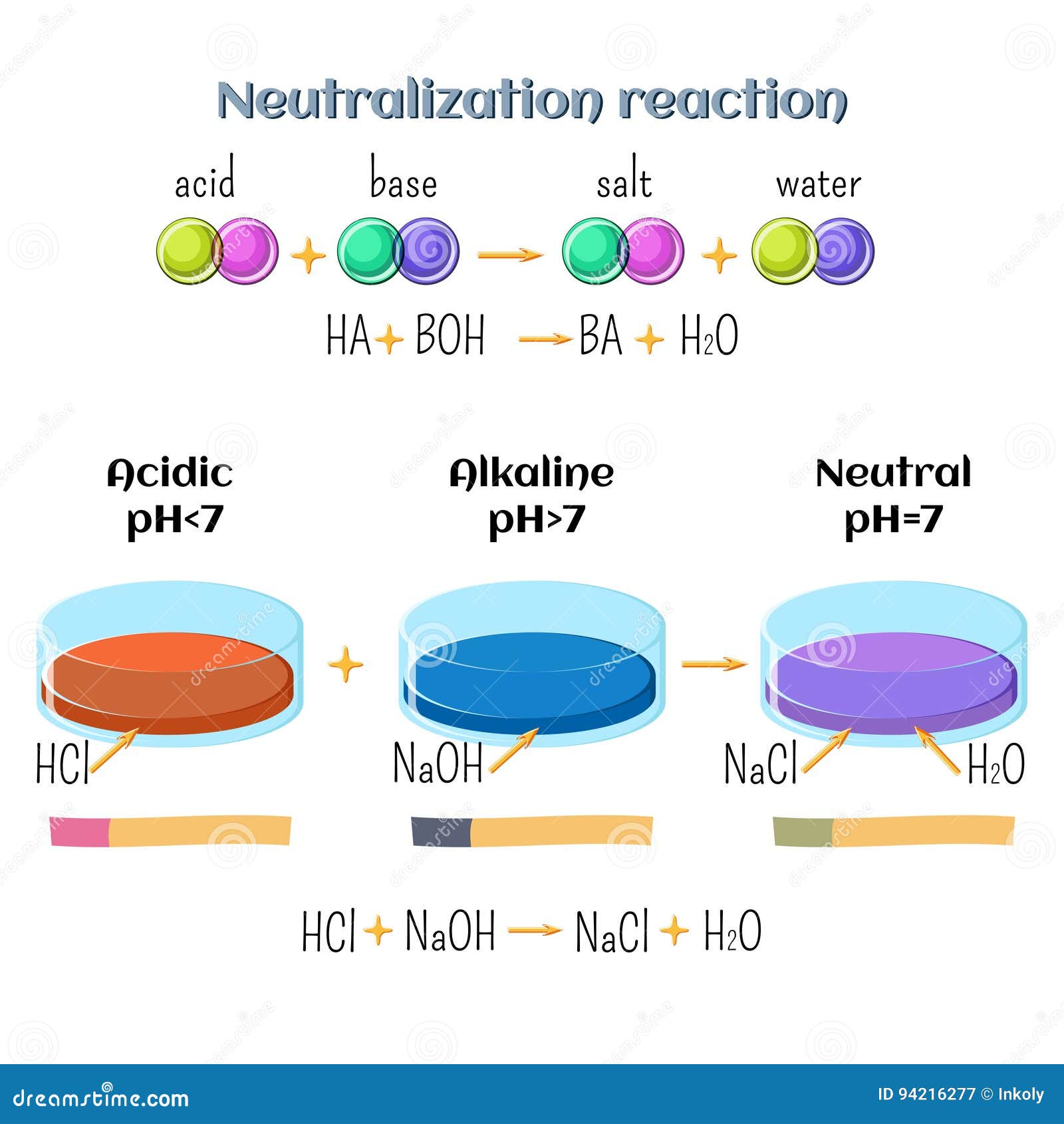
The Odd Couple: NaOH + HCl
Here's where things get interesting. When you mix Sodium Hydroxide and Hydrochloric Acid, they neutralize each other in a reaction. It's like watching two super-competitive siblings finally put aside their differences and work together. They create… table salt (NaCl) and water (H2O)! Yes, the very same salt you sprinkle on your fries. Mind. Blown.
The chemical equation looks like this: NaOH + HCl → NaCl + H2O. Simple, right? It's a classic acid-base reaction. The acid (HCl) donates a proton (H+) to the base (NaOH), and boom – neutrality achieved. It’s chemistry's way of saying, "Can't we all just get along?"

This neutralization reaction is also exothermic, meaning it releases heat. Ever mixed two chemicals together and noticed the container got warm? That’s likely an exothermic reaction at play! Be careful, because some reactions can get very hot. It's like when your siblings finally agree, but they're so enthusiastic about it that they almost set the house on fire with their newfound teamwork.
Why Should I Care?
So, why should you care about this seemingly obscure chemical reaction? Well, understanding acid-base reactions is fundamental to so many things! From understanding how antacids work (neutralizing excess stomach acid) to controlling the pH of your swimming pool (keeping it from becoming a swamp), these reactions are all around us. It's like understanding the rules of a sport – once you know them, you can appreciate the game much more.

Plus, knowing a little chemistry can make you feel like a total science wizard. Next time someone asks you about acids and bases, you can casually drop the knowledge that mixing Sodium Hydroxide and Hydrochloric Acid creates salt and water. Watch their jaws drop. Okay, maybe not, but you'll feel smarter, and that's what counts!
In short, Sodium Hydroxide and Hydrochloric Acid are like the unlikely heroes of the chemical world. They might sound scary, but they play crucial roles in our everyday lives, from keeping our drains clear to helping us digest food. And when they come together, they create something as simple and essential as salt. So, the next time you're salting your popcorn, remember the amazing chemistry that made it possible!