Sodium Bromide Molar Mass
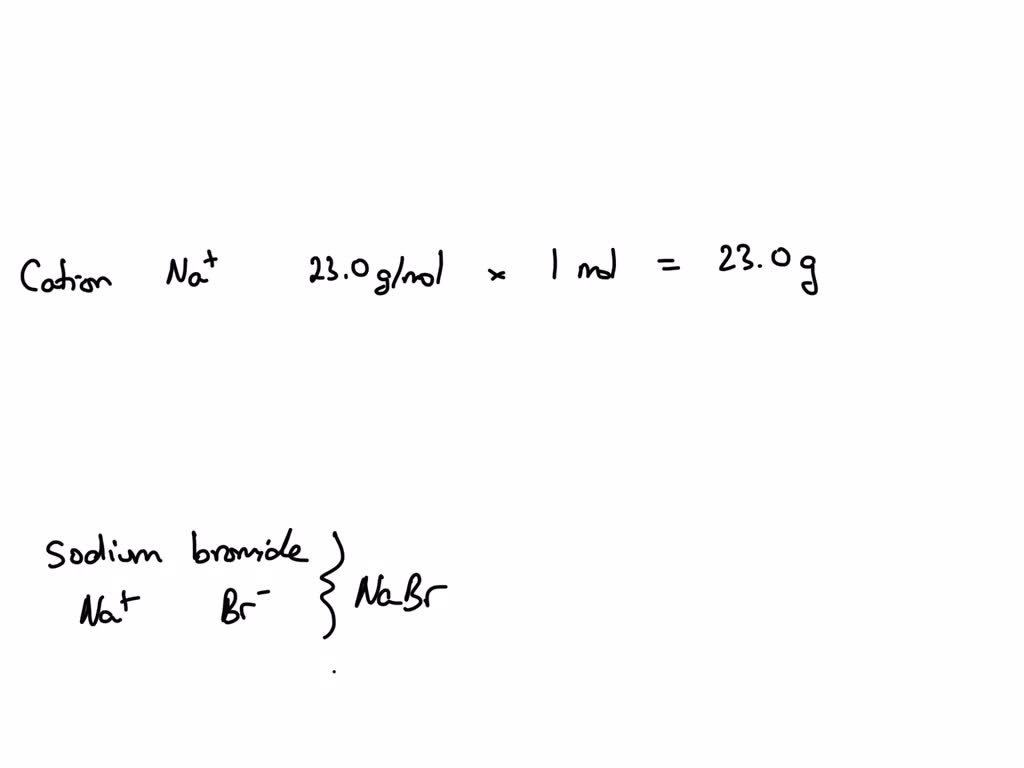
Okay, buckle up, buttercups! We're diving headfirst into the wonderfully weird world of…molar mass! Don't run away screaming! It sounds scary, but I promise it's easier than parallel parking on a Tuesday.
Today's star of the show? Sodium Bromide (NaBr)! Think of it as table salt’s slightly more dramatic cousin. (Table salt is Sodium Chloride). This stuff has some seriously cool applications, from helping in photography to… well, let’s just say it's been known to have a calming effect in certain situations. Wink wink.
Now, molar mass is just a fancy way of saying "how much does one mole of something weigh?" A mole is like a baker's dozen, except instead of donuts, we're talking about incredibly tiny things called molecules! Basically, we're figuring out the weight of a gigantic, pre-determined pile of Sodium Bromide.
Must Read
The Great Element Weigh-In
So, how do we find this magical number? Well, it's like making a super simple recipe. We just need to know the ingredients!
Our ingredients are Sodium (Na) and Bromine (Br). Sodium is a feisty alkali metal and Bromine is a somewhat stinky liquid.
Think of the periodic table as a giant grocery store for elements. Each element has its own special price tag (atomic weight) listed. It's usually found right underneath its symbol.
Let's hunt down those price tags. The atomic weight of Sodium (Na) is approximately 22.99 atomic mass units (amu). Think of amu as tiny, tiny grams. Bromine (Br) clocks in at roughly 79.90 amu. Not bad, right?
Okay, now the fun part! It's time to add 'em up! Because Sodium Bromide is one Sodium atom and one Bromine atom linked together, all we have to do is add their atomic weights.
The Math (Don’t Panic!)
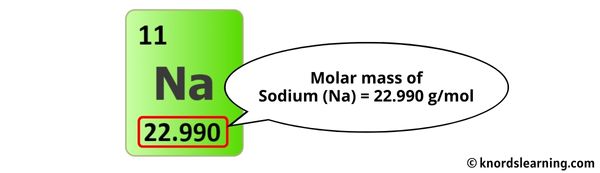
Ready? Brace yourselves! 22.99 (Sodium) + 79.90 (Bromine) = 102.89!
Voila! The molar mass of Sodium Bromide (NaBr) is approximately 102.89 grams per mole (g/mol). That means one mole of NaBr, which is a whopping 6.022 x 10^23 molecules (that’s a 6 followed by 23 zeros!), weighs about 102.89 grams. Pretty neat, huh?
Seriously, that number is so huge, it's practically unimaginable. If you had a mole of grains of sand, it would cover the entire surface of the Earth several feet deep! Glad we are dealing with Sodium Bromide.
Why Should You Care About Molar Mass?
Okay, so you might be thinking, "Great, I know the molar mass of Sodium Bromide. Now what?" Well, my friend, molar mass is actually super useful in all sorts of situations! It's like having a universal translator for chemical reactions.

Imagine you're a mad scientist (or a meticulous baker). You need a specific amount of Sodium Bromide for your latest experiment (or cake recipe). Knowing the molar mass allows you to convert between grams and moles. Meaning you can measure out exactly the right amount!
For example, let's say you need 0.5 moles of Sodium Bromide. You know that 1 mole weighs 102.89 grams. So, 0.5 moles would weigh half of that: 51.445 grams. BOOM! Precise measurements achieved!
Without molar mass, you'd be stuck guessing. And nobody wants to guess when it comes to chemical reactions (or baking). Unless you like explosions (or burnt cake).
"Molar mass: It's like a chemical GPS. It tells you exactly where you need to go!"
Chemists use molar mass to calculate the concentration of solutions, predict the amount of product formed in a reaction, and perform all sorts of other magical calculations. It's like the Swiss Army knife of chemistry!
Beyond Sodium Bromide: Molar Mass Mania
The fun doesn't stop with Sodium Bromide! You can calculate the molar mass of any compound, from water (H2O) to complicated organic molecules. Just find the atomic weights of each element in the compound and add 'em all up! Easy peasy!
For example, let's calculate the molar mass of water (H2O). Hydrogen (H) has an atomic weight of about 1.01 amu. Oxygen (O) has an atomic weight of about 16.00 amu.
Since there are two hydrogen atoms in water, we multiply hydrogen's atomic weight by 2: 1.01 x 2 = 2.02.
Then, we add that to the atomic weight of oxygen: 2.02 + 16.00 = 18.02 g/mol.
Ta-da! The molar mass of water is about 18.02 grams per mole. Keep that in mind next time you pour yourself a glass!
Molar Mass: Your New Best Friend (Maybe)
So, there you have it! The molar mass of Sodium Bromide (and a little extra molar mass goodness) demystified! It’s not so scary after all, is it?

Understanding molar mass can seem like a niche skill. However, you can impress your friends at parties and be the star of the chemistry club! Just kidding. Mostly.
But seriously, molar mass is a fundamental concept in chemistry, and hopefully, this little journey has made it a bit more accessible (and maybe even a little bit fun!). Now go forth and conquer the world of moles! Or at least understand them a little better.
Remember, with a little practice and a good periodic table, you can calculate the molar mass of anything! Now, if you'll excuse me, I'm off to calculate the molar mass of chocolate chip cookies… for science!
Consider this your official permission to geek out about molar mass at your next social gathering. Just don't blame me if everyone suddenly claims to have a prior engagement.
And that's a wrap! Now go forth and calculate the molar mass of all the things! Or, you know, just relax and enjoy a nice cup of tea. But if you do calculate the molar mass of tea… let me know what you get!
