Resonance Structures For N3-

Okay, buckle up buttercups, because we're diving headfirst into the wacky world of the azide ion, N3-! And trust me, it's way more exciting than it sounds (which, admittedly, isn't saying much at first glance).
Think of it like this: N3- is a molecule that's trying to decide which outfit to wear, but it can't quite make up its mind, so it wears them all at once! Sort of.
The Nitty-Gritty: Three Nitrogen Atoms, One Negative Charge
Basically, we're talking about three nitrogen atoms all chained together. Nitrogen, as you might recall, is a notorious show-off, always wanting to make three bonds.
Must Read
Now, throw in a negative charge into the mix, and things get interesting. This negative charge throws the normal bonding arrangement into a delightful state of chaos!
Picture This: Sharing is Caring (But Sometimes Confusing)
Imagine three kids sharing two candy bars. Sounds simple, right?
But what if those candy bars have to be distributed so that each kid gets at least a little bit? That’s the essence of what's going on with N3-. The bonds and the extra electrons are playing a game of musical chairs.
We use something called resonance structures to describe this bonding dilemma. They are just different ways of drawing the same molecule.
Resonance Structures: Like Multiple Personalities (of a Molecule)
A resonance structure is basically one possible arrangement of electrons and bonds in a molecule. We use arrows to show that the actual molecule is a hybrid or blend of all these arrangements.
It's like if you drew three different versions of the same cartoon character, each with slightly different features, but you knew they were all supposed to be the same character. They’re all contributing to the overall picture of what the molecule looks like.
With N3-, we can draw three major resonance structures. Brace yourselves, this is where the fun really begins!

Resonance Structure #1: N≡N+–N-
In this scenario, the first two nitrogen atoms form a triple bond, a very strong and stable connection. The first nitrogen has donated an electron and bears a positive charge and the last nitrogen has accepted an electron bearing a negative charge.
Think of it like two nitrogen atoms are super close friends and the other is like a distant acquaintance. This is a linear molecule and it's really hard to bend this structure.
It's a bold move, Nitrogen! Let's see if it pays off in the end.
Resonance Structure #2: N-–N+≡N
This is basically the mirror image of the first structure. The triple bond is now between the second and third nitrogen atoms, with the negative charge on the first nitrogen.
It’s like the first nitrogen and second are close while the third nitrogen is the distant acquaintance.
It’s the same story, just told from a different perspective. Two atoms form a triple bond and the other takes the charge.
Resonance Structure #3: N=N+=N-
Here, we get a more balanced distribution of electrons. Now, both nitrogen atoms are double bonded!
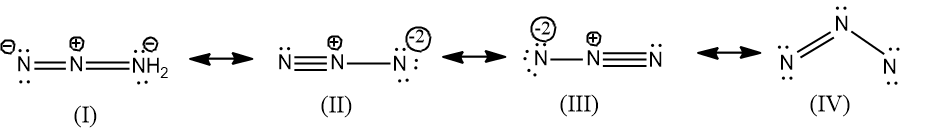
The central nitrogen atom is sporting a positive charge and the last nitrogen atom is bearing the negative charge.
Consider this a more “democratic” distribution of the electron love. This structure looks like it’s trying to make everyone happy!
The Resonance Hybrid: A Blending of Personalities
The real N3- ion isn't constantly flipping between these three structures. Oh no! The real N3- is a blend of all three at the same time. This is called the resonance hybrid.
Imagine mixing red, blue and yellow paint. You don't get a constantly flickering mixture of red, blue, and yellow. You get green!
The resonance hybrid is like the green paint of the molecular world. It's a single, stable thing with characteristics of all its contributors.
What This Means: Stability and Shared Responsibility
Resonance generally makes molecules more stable. By spreading out the electrons and charges, the molecule becomes less reactive and less likely to explode (a very good thing when dealing with azides!).
Think of it like spreading out the workload amongst a group of friends. If everyone pitches in, the job is easier and no one gets burned out. Spreading out the electrons makes the whole molecule more chill.
This is why resonance is such an important concept in chemistry. It helps us understand why certain molecules behave the way they do.

Why Do We Even Need Resonance Structures?
Because sometimes, a single Lewis structure just isn't enough to accurately describe the bonding in a molecule. It's like trying to describe a complex painting with just one color.
Resonance structures give us a more complete picture, showing us all the possible ways the electrons can be arranged. They also give us insights into the molecule's stability, reactivity, and other properties.
Basically, they are the secret decoder rings of the molecular world, helping us unlock the mysteries of chemical bonding!
A Real-World Analogy: The Team Effort
Think about a basketball team. Each player has their own strengths and weaknesses.
Sometimes one player is the star, scoring all the points. Other times, it's a team effort, with everyone contributing to the win. It’s kinda like sharing the electrons around the molecule.
The resonance structures of N3- are like different team formations, each emphasizing different players and their roles. The resonance hybrid is like the overall team strategy, incorporating the best aspects of each formation.
The Takeaway: Embrace the Ambiguity!
So, the next time you encounter resonance structures, don't panic! Embrace the ambiguity and remember that you're just looking at different perspectives of the same molecule.

They may seem confusing at first, but once you grasp the underlying concept, they become a powerful tool for understanding the fascinating world of chemistry.
And who knows, maybe you'll even start seeing resonance structures in your everyday life! (Okay, probably not, but it's fun to imagine, right?)
Bonus Fun Fact: Azides and Explosions!
Okay, I mentioned that azides can be explosive. In fact, sodium azide, NaN3, is used in car airbags. When the airbag is triggered, it decomposes rapidly, producing nitrogen gas which inflates the bag.
The stability afforded by resonance helps control the decomposition, but don't let that fool you. Under the right circumstances, azides can still pack a punch!
So, while we’re celebrating the wonders of resonance, let’s also appreciate the respect azides command. Remember: Handle with care!
In Conclusion: Resonance Rocks!
We've explored the world of N3- and its resonance structures. Hopefully, you now have a better understanding of this important concept in chemistry.
Resonance is all about shared responsibility, stability, and the beauty of multiple perspectives. It's a fundamental principle that helps us understand the behavior of molecules and the world around us.
So go forth and embrace the resonance! You're now one step closer to becoming a chemistry guru! You got this!
